EGF receptor in organ development, tissue homeostasis and regeneration
- PMID: 39966897
- PMCID: PMC11837477
- DOI: 10.1186/s12929-025-01119-9
EGF receptor in organ development, tissue homeostasis and regeneration
Abstract
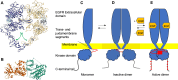
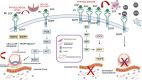
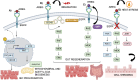
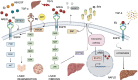
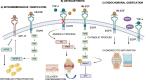
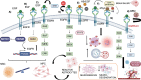
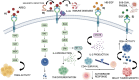
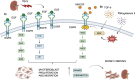
The epidermal growth factor receptor (EGFR) is a protein embedded in the outer membrane of epithelial and mesenchymal cells, bone cells, blood and immune cells, heart cells, glia and stem neural cells. It belongs to the ErbB family, which includes three other related proteins: HER2/ErbB2/c-neu, HER3/ErbB3, and HER4/ErbB4. EGFR binds to seven known signaling molecules, including epidermal growth factor (EGF) and transforming growth factor-alpha (TGF-α). This binding triggers the formation of receptor pairs (dimers), self-phosphorylation of EGFR, and the activation of several signaling pathways within the cell. These pathways influence various cellular processes like proliferation, differentiation, migration, and survival. EGFR plays a critical role in both development and tissue homeostasis, including tissue repair and adult organ regeneration. Altered expression of EGFR is linked to disruption of tissue homeostasis and various diseases, among which cancer. This review focuses on how EGFR contributes to the development of different organs like the placenta, gut, liver, bone, skin, brain, T cell regulation, pancreas, kidneys, mammary glands and lungs along with their associated pathologies. The involvement of EGFR in organ-specific branching morphogenesis process is also discussed. The level of EGFR activity and its impact vary across different organs. Factors as the affinity of its ligands, recycling or degradation processes, and transactivation by other proteins or environmental factors (such as heat stress and smoking) play a role in regulating EGFR activity. Understanding EGFR's role and regulatory mechanisms holds promise for developing targeted therapeutic strategies.
Keywords: EGF ligands; EGFR; Mammalian development; Signaling; Tissue homeostasis.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Ethics approval and consent to participate: Not applicable. Consent for publication: Not applicable. Competing interests: The authors declare that they have no competing interests.
Figures





References
-
- Cohen S. Epidermal growth factor (nobel lecture). Angew Chemie Int Ed English. 1987;26:717–22.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

