Real-world Outcomes Following Biologic Initiation in US Patients with Chronic Rhinosinusitis with Nasal Polyps
- PMID: 39969781
- PMCID: PMC11929629
- DOI: 10.1007/s12325-025-03120-y
Real-world Outcomes Following Biologic Initiation in US Patients with Chronic Rhinosinusitis with Nasal Polyps
Abstract
Introduction: Chronic rhinosinusitis with nasal polyposis (CRSwNP) is often associated with persistent symptoms and impaired quality of life despite treatment with intranasal corticosteroids. Biologics (dupilumab, mepolizumab, omalizumab) have been recently approved for CRSwNP. This study aims to characterize biologic use and real-world outcomes, including medication use and nasal polyps (NP) surgeries, following biologic treatment in US patients with CRSwNP.
Methods: This retrospective cohort study analyzed linked data from IQVIA longitudinal prescription and medical claims databases (July 2018-June 2023). Patients evaluated included those with ≥ 2 diagnoses of CRSwNP and ≥ 12 months of baseline data (overall cohort, index = first observed CRSwNP diagnosis) as well as patients with CRSwNP who received ≥ 2 consecutive biologic doses and had ≥ 24 months of follow-up data (biologic cohort, index = first biologic).
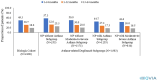
Results: Of 74,480 patients with CRSwNP, 8716 (12.0%) received a biologic and 2208 met all inclusion criteria. Dupilumab was the most frequently received biologic (89.8%; mepolizumab, 5.3%; omalizumab, 4.8%). Relative to the overall cohort, the biologic cohort was younger (mean age: 52.6 vs. 57.6 years), had more women (54.0% vs. 46.1%) and had a higher baseline prevalence of asthma (72.4% vs. 30.9%), allergic rhinitis (70.6% vs. 37.4%), NP surgery (15.8% vs. 5.8%), oral corticosteroid (OCS) use (84.0% vs. 51.8%), and antibiotic use (84.2% vs. 68.7%). During the 24 months after biologic initiation, 65.6% of patients had ≥ 1 OCS use (≥ 2 OCS uses during months 1-12, 27.0%; during months 13-24, 27.0%) and 77.9% had ≥ 1 antibiotic use; and 7.1% of patients without NP surgery before biologic initiation had ≥ 1 NP surgery during follow-up. Almost half of patients (49.3%) discontinued (≥ 90 days without receipt) their initial biologic during follow-up.
Conclusion: Biologic use was relatively low among US patients with CRSwNP. OCS and antibiotic usage among patients with CRSwNP remained substantial despite use of currently approved biologics, indicating an unmet need for improved treatment options.
Keywords: Allergist/immunologist; Biologics; Chronic rhinosinusitis with nasal polyposis (CRSwNP); Dupilumab; Ear-nose-throat; Mepolizumab; Omalizumab; Otolaryngologist; Real-world data.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Conflict of Interest: Anju T. Peters has received fees for sponsored research from AstraZeneca, Merck and Sanofi, and consultancy fees from AstraZeneca, Chiesi, GSK, Merck, Eli Lilly, and Sanofi. Joseph K. Han has received consultancy fees from AstraZeneca, Genentech, Gossamer Bio, GSK, Novartis, Regeneron Pharmaceuticals and Sanofi. Joseph D. Spahn, Sze-jung S. Wu, Tham T Le, and Christopher S. Ambrose are employees of AstraZeneca and may own stock or stock options in AstraZeneca. Andrew W. Lindsley is an employee of Amgen and owns stock in Amgen. Rifat Tuly, Elizabeth J. Wang, Xiaohui Zhao and Aimee M. Near are employees of IQVIA, which received funding from AstraZeneca to conduct the research study. Inyoung Lee was an employee of IQVIA during the study conduct and is currently employed by AbbVie. Ethical Approval: As a retrospective study using secondary data, no interventions were made to patients during this study. In compliance with the Health Insurance Portability and Accountability Act (HIPAA), patient data included in the analyses were de-identified; therefore, this study was exempt from Institutional Review Board (IRB) review. Permission was obtained to access and use the data from the IQVIA longitudinal prescription (LRx) and medical claims (Dx) databases, which are owned by IQVIA.
Figures




References
-
- Chen S, Zhou A, Emmanuel B, Thomas K, Guiang H. Systematic literature review of the epidemiology and clinical burden of chronic rhinosinusitis with nasal polyposis. Curr Med Res Opin. 2020;36(11):1897–911. - PubMed
-
- Mullol J, Azar A, Buchheit KM, Hopkins C, Bernstein JA. Chronic rhinosinusitis with nasal polyps: quality of life in the biologics era. J Allergy Clin Immunol Pract. 2022;10(6):1434–53. - PubMed
-
- Thomas WW 3rd, Harvey RJ, Rudmik L, Hwang PH, Schlosser RJ. Distribution of topical agents to the paranasal sinuses: an evidence-based review with recommendations. Int Forum Allergy Rhinol. 2013;3(9):691–703. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

