Viral reactivations and fungal infections in nonresolving acute respiratory distress syndrome
- PMID: 39971398
- PMCID: PMC11836671
- DOI: 10.1183/16000617.0153-2024
Viral reactivations and fungal infections in nonresolving acute respiratory distress syndrome
Abstract
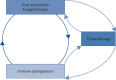
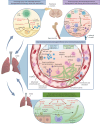
Acute respiratory distress syndrome (ARDS) is a condition affecting 10% of patients requiring admission to the intensive care unit and results from endothelial dysfunction, alveolar epithelial injury and unbalanced inflammation, leading to exudative pulmonary oedema. A significant portion of these patients experience a lung injury that fails to resolve. Persistent or worsening respiratory failure beyond 5 days after the initiation of mechanical ventilation is referred to as nonresolving ARDS. Viral and fungal pathogens can exploit the hyperinflammatory environment and altered immune landscape in ARDS, perpetuating a cycle of ongoing inflammation and lung injury, thereby contributing to the progression towards and persistence of nonresolving ARDS, even in previously immunocompetent patients. This review discusses the significance, pathophysiology, diagnostic challenges and key knowledge gaps concerning various viral and fungal pathogens in nonresolving ARDS, with a particular focus on influenza-associated and COVID-19-associated pulmonary aspergillosis and pulmonary reactivation of Herpesviridae, such as cytomegalovirus and herpes simplex virus. Diagnosing these infections is challenging due to their nonspecific clinical presentation and the inability of current tests to distinguish between fungal colonisation or asymptomatic viral shedding and clinically significant infections or reactivations. A deeper understanding of the complex interplay between these pathogens and the host immune system in the context of ARDS, combined with advances in diagnostic and therapeutic strategies, has the potential to enhance the management and prognosis of patients with nonresolving ARDS.
Copyright ©The authors 2025.
Conflict of interest statement
Conflict of interest: J. Heylen reports support for the present manuscript from FWO (fundamental research grant 11PBR24N) and has received support for conference attendance from Gilead. J. Wauters received an institutional research fund from Pfizer; received investigator-initiated grants from Pfizer, Gilead, MSD, European Union's Horizon 2020 research and innovation programme (under grant agreement no 847507 HDM-FUN) and FWO project funding (under Grant G053121N); received speakers’ and travel fees from Pfizer, Gilead and MSD; declares participation in advisory boards for Pfizer and Gilead; and declares receipt of study drugs from MSD. L.D.J. Bos received an institutional research fund from Longfonds, IMI, Amsterdam UMC, ZonMW, Volition and Santhera; declares participation in advisory boards for Sobi, Impentri, Novartis, AstraZeneca and CSL Behring; consultancy fees from Scailyte; and declares participation in a DSMB for Aptarion. S. Feys reports support for the present manuscript from FWO (PhD fellowship (11M6922N and 11M6924N)), has received support for conference attendance from Pfizer and Gilead, support for a public PhD defense from Pfizer, Gilead and Mundipharma, and declares receipt of a speaker's fee from The Healthbook Company. The other authors declare no conflict of interest.
Figures
References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical
