Human urine stem cells protect against cyclophosphamide-induced premature ovarian failure by inhibiting SLC1A4-mediated outflux of intracellular serine in ovarian granulosa cells
- PMID: 39972244
- PMCID: PMC11840982
- DOI: 10.1186/s11658-025-00701-1
Human urine stem cells protect against cyclophosphamide-induced premature ovarian failure by inhibiting SLC1A4-mediated outflux of intracellular serine in ovarian granulosa cells
Abstract
Background: Cyclophosphamide (CTX) is the first-line medication for the treatment of breast cancer, although it potentially leads to severe ovarian dysfunction and even premature ovarian failure (POF). However, the mechanism of CTX-induced POF remains unclear. Mesenchymal stem cell-based therapy has been wildly used for treating numerous diseases. Therefore, our study aims to elucidate the underlying mechanism of CTX-induced POF and to explore the therapeutic effect of human urine stem cells (hUSCs) in POF.
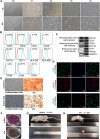
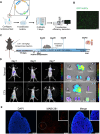
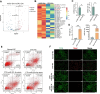
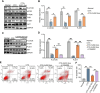
Methods: CTX-induced POF or ovarian granulosa cell (GCs) apoptosis were treated with hUSCs and their exosomes in vitro and in vivo. Morphological, histological, and functional alternations were examined using multiple approaches. The effector molecules of hUSC-derived exosomes (hUSC-Exo) were determined by differential expression analysis in the ovaries. The target genes of miRNA were accessed by transcriptome sequencing in GCs, and the underlying mechanisms were further elucidated.
Results: hUSCs remarkably inhibited CTX-induced apoptosis and promoted the proliferation of GCs, respectively. In addition, we observed that miR-27b-3p was highly expressed in hUSC-Exo and markedly suppressed CTX-induced GC apoptosis by specifically inhibiting the expression of SLC1A4, a serine transporter, in ovarian GCs, which, in turn, elevated the concentration of the intracellular serine by inhibiting the outflux of cellular serine. More importantly, the knockdown of SLC1A4 or simple supplementation of serine suppressed CTX-induced apoptosis of GCs. Finally, we demonstrated that CTX-induced apoptosis of ovarian GCs was essential for POF by reducing the intracellular serine concentration via elevating the expression of SLC1A4, whereas hUSCs protected against CTX-induced POF via miR-27b-3p/SLC1A4/serine axis-mediated activation of the PI3K/AKT/mTOR signaling pathway.
Conclusions: Our study suggests that hUSC-based cell therapy or simple supplementation of serine may provide an efficient therapeutic approach for the prevention and treatment of CTX-induced POF clinically.
Keywords: Cyclophosphamide; Human urine-derived stem cells; Ovarian granulosa cells; Premature ovarian failure; SLC1A4; miR-27b-3p.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Ethics approval and consent to participate: The study was conducted following the Declaration of Helsinki (as revised in 2013), and written consent for tissue donation was obtained from each patient. The protocol was approved by the Institutional Review Board of the First Affiliated Hospital of Nanchang University (approval no. 2023CDYFYYLK08-015; approval date 11 August 2023). The protocols were approved by the Animal Research Ethics Board of Nanchang University (approval no. 20X045; approval date 2 July 2020), following the Guidelines for the Care and Use of Animals. The study involving animals was conducted following the Basel Declaration. Consent for publication: Not applicable. Competing interests: The authors declare no competing interests.
Figures




References
-
- van Hellemond IEG, Vriens IJH, Peer PGM, Swinkels ACP, Smorenburg CH, Seynaeve CM, et al. Ovarian function recovery during anastrozole in breast cancer patients with chemotherapy-induced ovarian function failure. J Natl Cancer Inst. 2017. 10.1093/jnci/djx074. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

