Engineered Extracellular Vesicles as a New Class of Nanomedicine
- PMID: 39975802
- PMCID: PMC11835263
- DOI: 10.1021/cbe.4c00122
Engineered Extracellular Vesicles as a New Class of Nanomedicine
Abstract
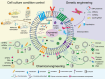
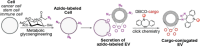
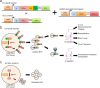
Extracellular vesicles (EVs) are secreted from biological cells and contain many molecules with diagnostic values or therapeutic functions. There has been great interest in academic and industrial communities to utilize EVs as tools for diagnosis or therapeutics. In addition, EVs can also serve as delivery vehicles for therapeutic molecules. An indicator of the enormous interest in EVs is the large number of review articles published on EVs, with the focus ranging from their biology to their applications. An emerging trend in EV research is to produce and utilize "engineered EVs", which are essentially the enhanced version of EVs. EV engineering can be conducted by cell culture condition control, genetic engineering, or chemical engineering. Given their nanometer-scale sizes and therapeutic potentials, engineered EVs are an emerging class of nanomedicines. So far, an overwhelming majority of the research on engineered EVs is preclinical studies; there are only a very small number of reported clinical trials. This Review focuses on engineered EVs, with a more specific focus being their applications in therapeutics. The various approaches to producing engineered EVs and their applications in various diseases are reviewed. Furthermore, in vivo imaging of EVs, the mechanistic understandings, and the clinical translation aspects are discussed. The discussion is primarily on preclinical studies while briefly mentioning the clinical trials. With continued interdisciplinary research efforts from biologists, pharmacists, physicians, bioengineers, and chemical engineers, engineered EVs could become a powerful solution for many major diseases such as neurological, immunological, and cardiovascular diseases.
© 2024 The Authors. Co-published by Zhejiang University and American Chemical Society.
Conflict of interest statement
The authors declare no competing financial interest.
Figures



Similar articles
-
Engineered Extracellular Vesicles: Emerging Therapeutic Strategies for Translational Applications.Int J Mol Sci. 2023 Oct 15;24(20):15206. doi: 10.3390/ijms242015206. Int J Mol Sci. 2023. PMID: 37894887 Free PMC article. Review.
-
Targeted delivery of extracellular vesicles: the mechanisms, techniques and therapeutic applications.Mol Biomed. 2024 Nov 21;5(1):60. doi: 10.1186/s43556-024-00230-x. Mol Biomed. 2024. PMID: 39567444 Free PMC article. Review.
-
Native and engineered extracellular vesicles for wound healing.Front Bioeng Biotechnol. 2022 Dec 9;10:1053217. doi: 10.3389/fbioe.2022.1053217. eCollection 2022. Front Bioeng Biotechnol. 2022. PMID: 36568307 Free PMC article. Review.
-
Biomimicking Extracellular Vesicles with Fully Artificial Ones: A Rational Design of EV-BIOMIMETICS toward Effective Theranostic Tools in Nanomedicine.ACS Biomater Sci Eng. 2023 Nov 13;9(11):5924-5932. doi: 10.1021/acsbiomaterials.2c01025. Epub 2022 Dec 19. ACS Biomater Sci Eng. 2023. PMID: 36535896 Free PMC article. Review.
-
New insights of engineered extracellular vesicles as promising therapeutic systems.Extracell Vesicles Circ Nucl Acids. 2023 Apr 27;4(2):191-194. doi: 10.20517/evcna.2023.22. eCollection 2023. Extracell Vesicles Circ Nucl Acids. 2023. PMID: 39697986 Free PMC article.
Cited by
-
Hydroxychloroquine-Derived Ionizable Lipid Facilitates Spleen-Tropic Transfection and Enhances Cancer Immunotherapy.Chem Bio Eng. 2025 Jun 18;2(7):437-448. doi: 10.1021/cbe.5c00031. eCollection 2025 Jul 24. Chem Bio Eng. 2025. PMID: 40735013 Free PMC article.
References
-
- Skog J.; Würdinger T.; van Rijn S.; Meijer D. H.; Gainche L.; Curry W. T. Jr; Carter B. S.; Krichevsky A. M.; Breakefield X. O. Glioblastoma Microvesicles Transport RNA and Proteins that Promote Tumour Growth and Provide Diagnostic Biomarkers. Nat. Cell. Bio. 2008, 10, 1470–1476. 10.1038/ncb1800. - DOI - PMC - PubMed
Publication types
LinkOut - more resources
Full Text Sources
