Targeting Histone Deacetylase 11 with a Highly Selective Inhibitor for the Treatment of MASLD
- PMID: 39976110
- PMCID: PMC12005767
- DOI: 10.1002/advs.202412903
Targeting Histone Deacetylase 11 with a Highly Selective Inhibitor for the Treatment of MASLD
Abstract
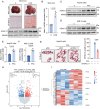
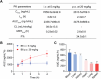
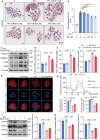
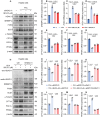
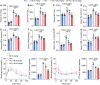
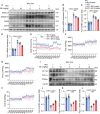
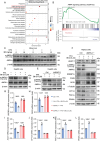
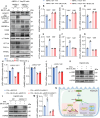
Metabolic dysfunction-associated steatotic liver disease (MASLD) represents the most prevalent chronic liver disorder globally. Due to its intricate pathogenesis and the current lack of efficacious pharmacological interventions, there is a pressing need to discover novel therapeutic targets and agents for MASLD treatment. Herein, it is found that histone deacetylase 11 (HDAC11), a subtype of HDAC family, is markedly overexpressed in both in vitro and in vivo models of MASLD. Furthermore, the knockdown of HDAC11 is observed to mitigate lipid accumulation in hepatic cells. A highly selective HDAC11 inhibitor, B6, which exhibits favorable pharmacokinetic property and liver distribution, is further designed and synthesized. Integrating RNA-seq data with in vivo and in vitro experiments, B6 is found to inhibit de novo lipogenesis (DNL) and promote fatty acid oxidation, thus mitigating hepatic lipid accumulation and pathological symptoms in MASLD mice. Further omics analysis and experiments reveal that B6 enhances the phosphorylation of AMPKα1 at Thr172 through the inhibition of HDAC11, consequently modulating DNL and fatty acid oxidation in the liver. In summary, this study identifies HDAC11 as a potential therapeutic target in MASLD and reports the discovery of a highly selective HDAC11 inhibitor with favorable drug-like properties for the treatment of MASLD.
Keywords: AMP‐activated protein kinase; de novo lipogenesis; fatty acid oxidation; histone deacetylase 11; metabolic dysfunction‐associated steatotic liver disease.
© 2025 The Author(s). Advanced Science published by Wiley‐VCH GmbH.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



Similar articles
-
Polyoxometalates Ameliorate Metabolic Dysfunction-Associated Steatotic Liver Disease by Activating the AMPK Signaling Pathway.Int J Nanomedicine. 2024 Oct 25;19:10839-10856. doi: 10.2147/IJN.S485084. eCollection 2024. Int J Nanomedicine. 2024. PMID: 39479173 Free PMC article.
-
Alpha-aminobutyric acid ameliorates diet-induced metabolic dysfunction-associated steatotic liver disease (MASLD) progression in mice via enhancing AMPK/SIRT1 pathway and modulating the gut-liver axis.J Nutr Biochem. 2025 Jun;140:109885. doi: 10.1016/j.jnutbio.2025.109885. Epub 2025 Feb 25. J Nutr Biochem. 2025. PMID: 40015656
-
Targeting acetyl-CoA carboxylases for the treatment of MASLD.J Lipid Res. 2024 Dec;65(12):100676. doi: 10.1016/j.jlr.2024.100676. Epub 2024 Oct 25. J Lipid Res. 2024. PMID: 39461620 Free PMC article. Review.
-
Circular RNA RRM2 alleviates metabolic dysfunction-associated steatotic liver disease by targeting miR-142-5p to increase NRG1 expression.Am J Physiol Gastrointest Liver Physiol. 2024 Oct 1;327(4):G485-G498. doi: 10.1152/ajpgi.00255.2023. Epub 2024 Jul 23. Am J Physiol Gastrointest Liver Physiol. 2024. PMID: 39259911
-
Understanding the Link Between Sterol Regulatory Element Binding Protein (SREBPs) and Metabolic Dysfunction Associated Steatotic Liver Disease (MASLD).Curr Obes Rep. 2025 Apr 14;14(1):36. doi: 10.1007/s13679-025-00626-y. Curr Obes Rep. 2025. PMID: 40227546 Review.
References
-
- Younossi Z., Anstee Q. M., Marietti M., Hardy T., Henry L., Eslam M., George J., Bugianesi E., Nat. Rev. Gastroenterol. Hepatol. 2018, 15, 11. - PubMed
MeSH terms
Substances
Grants and funding
- tsqn202211296/Taishan Scholars Program of Shandong Province
- 82103983/National Natural Science Foundation of China
- U22A20272/National Natural Science Foundation of China
- 82173807/National Natural Science Foundation of China
- ZR2023YQ062/Fundamental Research Funds for the Central Universities-Ocean University of China and the Science Foundation for Excellent Young Scholars of Shandong Province
LinkOut - more resources
Full Text Sources
