Concomitant medications in patients with metastatic urothelial carcinoma receiving enfortumab vedotin: real-world data from the ARON-2EV study
- PMID: 39976819
- PMCID: PMC11842414
- DOI: 10.1007/s10585-025-10335-4
Concomitant medications in patients with metastatic urothelial carcinoma receiving enfortumab vedotin: real-world data from the ARON-2EV study
Abstract
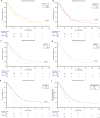
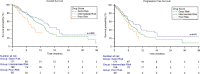
Patients with metastatic urothelial carcinoma (mUC) are typically elderly and often have other comorbidities that require the use of concomitant medications. In our study we evaluated the association of concomitant use of antibiotics (ATBs), proton pump inhibitors (PPIs), corticosteroids, statins, metformin and insulin with patient outcomes and we validated the prognostic role of a concomitant drug score in mUC patients treated with enfortumab vedotin (EV) monotherapy. Data from 436 patients enrolled in the ARON-2EV retrospective study were analyzed according to the concomitant medications used at baseline. Finally, the patients were stratified into three risk groups according to the concomitant drug score based on ATBs, corticosteroids and PPIs. Statistical analysis involved Fisher exact test, Kaplan-Meier method, log-rank test, and univariate/multivariate Cox proportional hazard regression models. Inferior survival outcomes were observed in ATB users compared to non-users (OS: 7.3 months, 95%CI 5.0 - 12.3 vs 13.7 months, 95%CI 12.2 - 47.3, p = 0.001; PFS: 5.1 months 95%CI 3.3 - 17.7 vs 8.3 months, 95%CI 7.1 - 47.3, p = 0.001) and also in corticosteroid users compared to non-users (OS: 8.4 months, 95%CI 6.6 - 10.0 vs 14.2 months, 95%CI 12.7 - 47.3, p < 0.001; PFS: 6.0 months 95%CI 4.6 - 7.9 vs 8.9 months, 95%CI 7.2 - 47.3, p = 0.004). In the Cox multivariate analysis, the concomitant drug score was a significant factor predicting both OS (HR = 1.32 [95% CI 1.03 - 1.68], p = 0.026) and PFS (HR = 1.23 [95% CI 1.01 - 1.51], p = 0.044). Our findings suggest detrimental impact of concomitant use of ATBs and corticosteroids on survival outcomes and the prognostic utility of the concomitant drug score in previously treated mUC patients receiving EV.
Keywords: ARON-2EV study; Antibiotics; Concomitant drug score; Concomitant medication; Corticosteroids; Enfortumab vedotin; NCT05290038; Urothelial cancer.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Conflict of interest: Ondrej Fiala received honoraria from Roche, Janssen, GSK and Pfizer for consultations and lectures unrelated to this project. Sebastiano Buti received honoraria as speaker at scientific events and advisory role by BMS, Pfizer, MSD, Ipsen, Roche, Eli Lilly, AstraZeneca, Pierre-Fabre, Novartis, Merck, Gentili, Astellas. All unrelated to the present paper. Alfonso Gómez de Liaño has received honoraria for advisory boards, consultation, or educational events from AAA HealthCare, Astellas, AstraZeneca, Bayer, BMS, Ipsen, Johnson & Johnson, MSD, Merck KGaA, Novartis, Recordati Rare Diseases and Roche (all unrelated to the present paper); and has Institutional research funding from AstraZeneca, Genmab A/S, Gilead Sciences, Johnson & Johnson, Merck KGaA, MSD, Roche and Syneos Health. Takahiro Kimura is a paid consultant/advisor of Astellas, AstraZeneca, Bayer, Janssen, Sanofi, and Takeda. Martin Angel received honoraria from Roche, Johnson & Johnson, Raffo and Pfizer for consultations and lectures unrelated to this project. Javier Molina-Cerrillo: reports research funding from Roche, Ipsen, Pfizer, and Janssen; travel support from Pfizer, Janssen, Ipsen, and BMS; a consulting or advisory role with Ipsen, Roche, BMS, Pfizer, Sanofi, Janssen, Astellas, Eisai, Adium and MSD. All unrelated to the present paper. Maria T Bourlon is a consultant of Bristol Myers Squibb, Merck, MSD, Gilead, Astellas and Asofarma and a speaker for Janssen Pharmaceuticasl, MSD, Merck, and Astellas. All unrelated to the present paper. Andrey Soares: Honoraria from Janssen, Pfizer, Bayer, AstraZeneca, Astellas Pharma, Merck Serono, Sanofi, Ipsen, Adium. Consulting or Advisory Role from Astellas Pharma, Janssen, Roche, Bayer, AstraZeneca, MSD, Bristol-Myers Squibb, Adium, Ipsen, Pfizer, Novartis. Research Funding from Bristol-Myers Squibb (Inst), Astellas (Inst), AstraZeneca (Inst). Travel, Accommodations, Expenses from Bayer, Janssen, Ipsen, Adium, MSD, Merck Serono. Ownership: BIO, Brazilian Information Oncology. All are unrelated to this study. Nonfinancial with no other potential conflicts of interest were reported. Yüksel Ürün has served on advisory board for Abdi-İbrahim, Astellas, AstraZeneca, Bristol Myers-Squibb, Deva, Eczacıbaşı, Gen ilaç, Gilead, GSK, Janssen, Merck, MSD, Novartis, Pfizer, Roche and received travel grants, honoraria or consultation fees from Abdi-İbrahim, Astellas, Bristol Myers-Squibb, Deva, Eczacıbaşı, Gen İlaç, Gilead, GSK, Janssen, Merck, Novartis, Pfizer and Roche. All unrelated to the present paper. Bohuslav Melichar has received honoraria from Roche, BMS, MSD, Novartis, Merck Serono, Eli Lilly, Pfizer, and AstraZeneca, all unrelated to the present paper. Alexandr Poprach has received payment or honoraria for lectures, presentations, speakers’ bureaus, manuscript writing, or educational events from BMS, Ipsen, Roche, Astellas, Merck, Eisai, MSD, Novartis, and Pfizer, unrelated to this project. Vincenza Conteduca has served as a consultant/advisory board member for Johnson&Johnson, Astellas, Merck, AstraZeneca, Amgen, EISAI, Recordati, Novartis, and Bayer and has received speaker honoraria or travel support from Astellas, Johnson&Johnson, Ipsen, Bayer, Gilead, and BristolMyers Squibb. Fernando Sabino M. Monteiro: Research support was provided by Merck Sharp Dome. Honoraria from Janssen, Ipsen, Bristol Myers Squibb, and Merck Sharp Dome. Ownership: BIO, Brazilian Information Oncology. All are unrelated to this study. Tomas Buchler has received payment or honoraria for lectures, presentations, speakers’ bureaus, manuscript writing, or educational events from Bristol Myers Squibb, Ipsen, Roche, Astellas, Merck, Eisai, Merck Sharp Dohme, Novartis, AstraZeneca, Janssen, and Pfizer, all unrelated to this project. Francesco Massari has received research support and/or honoraria from Advanced Accelerator Applications, Astellas, Astra Zeneca, Bayer, BMS, Janssen, Ipsen, MSD, Pfizer outside the submitted work. Javier Molina-Cerrillo declares consultant, advisory or speaker roles for IPSEN, Roche, Pfizer, Sanofi, Janssen, and BMS. JMC has received research grants from Pfizer, IPSEN and Roche. Shilpa Gupta is a consultant for Bristol Myers Squibb, Merck, Pfizer, Gilead, Bayer, Seattle Genetics, is speaker for Bristol Myers Squibb and has Institutional research funding from Seatte Genetics, Pfizer, Merck, Bristol Myers Squibb, Roche, Novartis, Tyra Biosciences. Matteo Santoni has received research support and honoraria from Janssen, Bristol Myers Squibb, Ipsen, MSD, Astellas and Bayer, all unrelated to the present paper. Other authors declare that they have no conflicts of interest that might be relevant to the contents of this manuscript. Ethical approval: The study protocol was approved on September 28, 2023, by the Ethical Committee of the coordinating center (Marche Region-Italy-No. 2022 39/7875, Study Protocol “ARON 2 Study” NCT05290038) and by the Institutional Review Boards of participating centers. Consent to participate: The Informed consent with subsequent analysis of the follow-up data was obtained from all participants. Consent for publication: All authors have approved the manuscript for publication.
Figures

References
-
- Mohanty SK, Lobo A, Cheng L (2023) The 2022 revision of the World Health Organization classification of tumors of the urinary system and male genital organs: advances and challenges. Human Pathol 1(136):123–143 - PubMed
-
- Rosenberg JE, Powles T, Sonpavde GP, Loriot Y, Duran I, Lee JL, Matsubara N, Vulsteke C, Castellano D, Mamtani R, Wu C, Matsangou M, Campbell M, Petrylak DP (2023) EV-301 long-term outcomes: 24-month findings from the phase III trial of enfortumab vedotin versus chemotherapy in patients with previously treated advanced urothelial carcinoma. Ann Oncol 34(11):1047–1054. 10.1016/j.annonc.2023.08.016 - PubMed
-
- Powles T, Bellmunt J, Comperat E, De Santis M, Huddart R, Loriot Y, Necchi A, Valderrama BP, Ravaud A, Shariat SF, Szabados B, van der Heijden MS, Gillessen S, ESMO Guidelines Committee (2024) ESMO clinical practice guideline interim update on first-line therapy in advanced urothelial carcinoma. Ann Oncol 35(6):485–490. 10.1016/j.annonc.2024.03.001 - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

