Temporal evolution of breast cancer brain metastases treatments and outcomes
- PMID: 39979277
- PMCID: PMC11842824
- DOI: 10.1038/s41523-025-00735-w
Temporal evolution of breast cancer brain metastases treatments and outcomes
Abstract
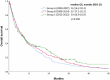
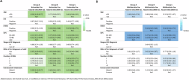
Brain metastases (BMs) are a common complication of advanced breast cancer (BC), and their management has significantly evolved. We evaluated the clinical impact of these changes dividing patients diagnosed with BCBMs at three Institutions according to year of BMs diagnosis: 2000-2007 (group A), 2008-2014 (group B) and 2015-2022 (group C). Stereotactic radiotherapy increased (p < 0.001), and WBRT decreased (p = 0.010) over time. Among HER2+ BC patients, more received anti-HER2 therapy after BM diagnosis in recent years (p < 0.011). Overall survival (OS) did not improve in the entire cohort (p = 0.260); however, OS improved in patients with HR-/HER2+ BC (median OS 8.7, 10.1, 23.7 months in group A, B, C, respectively; p = 0.002). HER2-positivity, not prognostic in group A, became prognostic in group C (p < 0.001). While therapy for patients with BCBMs significantly changed over two decades, an OS improvement was observed only in HR-/HER2+ patients, potentially due to increased availability of anti-HER2 therapies with intracranial activity.
© 2025. The Author(s).
Conflict of interest statement
Competing interests: MB reports travel support from Eli Lilly. GG reports fees for advisory role from Gilead, Seagen, Menarini, personal fees as an invited speaker from Eli Lilly, Novartis, MSD, travel support Gilead, Eli Lilly, Pfizer, Novartis, Daiichi Sankyo, AMGEN, AstraZeneca. SG reports honoraria and consulting fees from Pfizer, Lilly, Astra Zeneca and Gilead. CB reports travel expenses and fees for oral presentations and boards from Daiichi-Sankyo, Astra-zeneca, Seagen. FM reports personal fees from Roche, Novartis, Pfizer, Seagen, Menarini, MSD, Gilead, Astrazeneca. CZ reports travel support from Eli Lilly, Sanofi, Gilead. FG reports travel support from Eli Lilly, Gilead, Novartis; honoraria for lectures: AstraZeneca, Eli Lilly, Gilead. WJ reports grants, personal fees and non-financial support from Astra Zeneca, personal fees and non-financial support from Eisai, Novartis, Roche, Pfizer, Eli Lilly, Chugai and Gilead, personal fees from MSD, BMS and Seagen, grants and personal fees from Daiichi Sankyo, outside the submitted work. MVD reports personal fees for consultancy/advisory role from: Eli Lilly, Pfizer, Novartis, Seagen, Gilead, MSD, Exact Sciences, AstraZeneca, Roche, Daiichi Sankyo, Roche. AD reports honoraria for advisory board participation from Novocure, Servier Pharmaceuticals; travel grant from Servier Pharmaceuticals. VG reports personal fees for advisory board membership for AstraZeneca, Daiichi Sankyo, Eli Lilly, Gilead, MSD, Novartis, Pfizer, Olema Oncology, Pierre Fabre, Roche, Menarini Stemline, personal fees as an invited speaker for AstraZeneca, Daiichi Sankyo, Eli Lilly, Exact Sciences, Gilead, GSK, Novartis, Roche, Zentiva, Menarini Stemline, personal fees for expert testimony for Eli Lilly. The other authors declare no conflicts of interest.
Figures

References
-
- Globocan 2020, International Agency for Research on Cancer n.d. 10.8.
-
- Malvezzi M. et al. European Cancer Mortality Predictions For The Year 2023 With Focus On Lung Cancer. Ann Oncol 2023. 10.1016/j.annonc.2023.01.010. - PubMed
-
- Lin, N. U., Gaspar, L. E. & Soffietti, R. Breast Cancer in the Central Nervous System: Multidisciplinary Considerations and Management. Am. Soc. Clin. Oncol. Educ. Book37, 45–56 (2017). - PubMed
-
- Le Rhun, E. et al. EANO-ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up of patients with brain metastasis from solid tumours. Ann. Oncol.32, 1332–1347 (2021). - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

