Efficacy and Safety of Once-Daily Roflumilast Cream 0.05% in Pediatric Patients Aged 2-5 Years With Mild-to-Moderate Atopic Dermatitis (INTEGUMENT-PED): A Phase 3 Randomized Controlled Trial
- PMID: 39980188
- PMCID: PMC11950803
- DOI: 10.1111/pde.15840
Efficacy and Safety of Once-Daily Roflumilast Cream 0.05% in Pediatric Patients Aged 2-5 Years With Mild-to-Moderate Atopic Dermatitis (INTEGUMENT-PED): A Phase 3 Randomized Controlled Trial
Abstract
Background/objectives: Efficacy and safety of roflumilast cream 0.15% were demonstrated in patients aged ≥ 6 years with atopic dermatitis (AD) in two Phase 3 trials. This Phase 3 parallel-group, double-blind trial (INTEGUMENT-PED; NCT04845620) compared the efficacy and safety of roflumilast cream 0.05% and a vehicle in patients aged 2-5 years with AD.
Methods: Patients aged 2-5 years with mild-to-moderate AD were treated with once-daily roflumilast cream 0.05% or vehicle for 4 weeks. The primary efficacy endpoint was Validated Investigator Global Assessment for AD (vIGA-AD) Success (0 [Clear] or 1 [Almost Clear] plus ≥ 2-grade improvement from baseline) at Week 4. Other endpoints included ≥ 75% improvement in Eczema Area and Severity Index (EASI-75) and Worst Itch-Numeric Rating Score (WI-NRS) Success (≥ 4-point improvement in patients with baseline ≥ 4). Safety and tolerability were also assessed.
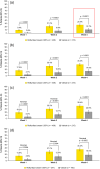
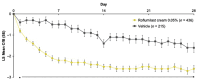
Results: Among 437 and 215 patients treated with roflumilast and vehicle, respectively, significantly greater proportions of the roflumilast group achieved Week-4 vIGA-AD Success (25.4% vs. 10.7%; p < 0.0001), EASI-75 (39.4% vs. 20.6%; p < 0.0001), and WI-NRS Success (35.3% vs. 18.0%; nominal p = 0.0002). Improvement in pruritus was observed within 24 h after the first application (nominal p = 0.0014). Treatment-emergent adverse event (TEAE) rates were low in both groups, and 98.9% were mild or moderate. At all timepoints, stinging/burning that caused definite discomfort was reported by ≤ 0.7% of caregivers of patients in the roflumilast group.
Conclusions: In this Phase 3 trial, once-daily roflumilast cream 0.05% improved AD signs/symptoms in patients aged 2-5 years, with early pruritus improvement, low AE rates, and local tolerability comparable with vehicle.
Trial registration: ClinicalTrials.gov: NCT04845620.
Keywords: atopic dermatitis; eczema; nonsteroidal topical; pediatrics; pruritus; roflumilast.
© 2024 The Author(s). Pediatric Dermatology published by Wiley Periodicals LLC.
Conflict of interest statement
Lawrence F. Eichenfield has been an investigator for AbbVie, Amgen, Arcutis, Bausch Health, Castle Biosciences, Dermavant, Galderma, Incyte, Pfizer, Regeneron, Sanofi‐Genzyme, and Target RWE; a consultant with an honorarium for AbbVie, Amgen, Apogee Therapeutics, Attovia, Arcutis, ASLAN Pharmaceuticals, BMS, Dermavant, Forte Biosciences, Galderma, Incyte, LEO Pharma, Lilly, Novartis, Ortho Dermatologics, Pfizer, Regeneron, and Sanofi‐Genzyme; and a board of directors member for Forte Biosciences. Rocco Serrao has been an investigator for AbbVie, Acelyrin, AnaptysBio, Arcutis, Concert Pharmaceuticals, Dermavant, Formation Bio, Lilly, and Regeneron; served on speaker's bureaus for Dermavant, Incyte, LEO Pharma, Lilly, Pfizer, Regeneron, and Sanofi; and on advisory boards for AbbVie, Arcutis, Dermavant, Pfizer, and Regeneron. Vimal H. Prajapati has been an advisor, consultant, and/or speaker for AbbVie, Actelion, Amgen, Apogee Therapeutics, Aralez Bio, Arcutis, Aspen Pharmacare, Bausch Health, BioScript Solutions, Boehringer Ingelheim, BMS, Celgene, Celltrion, Cipher Pharmaceuticals, CorEvitas, Galderma, GSK, Homeocan, Incyte, JAMP Pharma, Janssen, Johnson & Johnson, Johnson & Johnson Innovative Medicine, LEO Pharma, Lilly, Medexus, Novartis, Organon, Pediapharm, Pfizer, Regeneron, Sanofi‐Genzyme, Sun Pharma, Tribute Pharmaceuticals, UCB, and Valeant; investigator for AbbVie, AnaptysBio, Apogee Therapeutics, Arcutis, Arena Pharmaceuticals, Asana Biosciences, Bausch Health, Boehringer Ingelheim, BMS, Celgene, Concert Pharmaceuticals, CorEvitas, Dermavant, Dermira, Galderma, Incyte, Janssen, LEO Pharma, Lilly, Meiji Seika Pharma, Nektar Therapeutics, Nimbus Lakshmi, Novartis, Pfizer, RAPT Therapeutics, Regeneron, Reistone, Sanofi‐Genzyme, Sun Pharma, Takeda, and UCB; and received grants from AbbVie, Bausch Health, Janssen, LEO Pharma, Novartis, and Sanofi‐Genzyme. John C. Browning has been an investigator for AbbVie, Arcutis, Aslan, Concert Pharmaceuticals, Galderma, GSK, Pfizer, Regeneron, Sanofi, and UCB; a consultant with an honorarium for Dermavant, Krystal Biotech, Pfizer, and Regeneron; and chair of data safety monitoring board for Verricadermics and member of data safety monitoring board for Abeona. Lisa Swanson has been an investigator for Arcutis and a consultant with honorarium for AbbVie, Arcutis, Castle Biosciences, Dermavant, Incyte, LEO Pharma, Lilly, Pfizer, Regeneron, and Sanofi. Tracy Funk has been an investigator for AbbVie, Amgen, Arcutis, and Palvella Therapeutics; a consultant with an honorarium for Bain Capital, Guidepoint, Palvella Therapeutics, and Sanofi; and an advisory board participant for Arcutis and Incyte. Mercedes E. Gonzalez has been an investigator for AbbVie, Arcutis, Amgen, Anterogen, Dermavant, Incyte, Krystal Biotech, Lilly, Neilsen Biosciences, Novartis, Pfizer, Regeneron, and Sanofi; a consultant with an honorarium for Abeona, Arcutis, Alphyn Biologics, Apogee Therapeutics, Dermavant, Galderma, Incyte, Krystal Biotech, LEO Pharma, Lilly, Pelthos Therapeutics, and Verrica; and on speakers bureaus for AbbVie, Krystal Biotech, LEO Pharma, Lilly, Pfizer, Regeneron, Sanofi‐Genzyme, and Verrica. Adelaide A. Hebert has received research grants paid to UTHealth McGovern Medical School‐Houston from Arcutis, Brickell Biotech, Dermavant, Galderma, LEO Pharma, Lilly, Mayne Pharma, Ortho Dermatologics, and Pfizer; has received honoraria from Almirall, Arcutis, Bristol‐Myers Squibb, Dermavant, Galderma, Incyte, Janssen, LEO Pharma, Lilly, Mayne Pharma, Ortho Dermatologics, Pfizer, Verrica, and Novan Therapeutics; and serves on data safety monitoring boards for Bausch, GSK, Regeneron, and Sanofi. Mark Lee has been an investigator for AbbVie, Alumis, Arcutis, Bluefin Biomedicine, Cara Therapeutics, Dermavant, LEO Pharma, Merck, Pfizer, and RAPT Therapeutics. Mark Boguniewicz has been an investigator for Incyte, Regeneron, Sanofi, and advisory boards for AbbVie, Amgen, Arcutis, ASLAN Pharmaceuticals, Dermavant, Eli Lilly, Glaxo, LEO Pharma, Pfizer, Regeneron, Sanofi‐Genzyme. Eric L. Simpson has been an investigator for AbbVie, Amgen, Arcotech Biopharma, Arcutis, ASLAN Pharmaceuticals, Castle Biosciences, CorEveitas, Dermavant, Dermira, Galderma, Incyte, Kymab, Kyowa Hakko Kirin Pharma, LEO Pharma, Lilly, Merck, National Jewish Health, Novartis, Pfizer, Regeneron, Sanofi, Target RWE, Tioga Pharmaceuticals, and Vanda Pharmaceuticals; a consultant with honorarium for AbbVie, Advances in Cosmetic Medical Derm Hawaii LLC, Amgen, AOBiome, Arcutis, Arena Pharmaceuticals, ASLAN Pharmaceuticals, BenevolentAI, BiomX, Bluefin Biomedicine, Boehringer Ingelheim, Boston Consulting Group, Bristol‐Myers Squibb, Collective Acumen, CorEvitas, Coronado Biosciences, Demira, Evidera, Evelo Biosciences, Excerpta Medica, Forte Biosciences, Gesellschaft Z, GSK, Incyte, Janssen, Johnson & Johnson, Kyowa Hakko Kirin Pharma, LEO Pharma, Lilly, Med Learning Group, Medscape, MJH Holding Company, Ortho Dermatologics, Pfizer, Physicians World LLC, Pierre Fabre, Regeneron, Roivant, Sanofi, Trevi Therapeutics, Valeant, and WebMD; on speaker bureaus for FIDE, Maui Derm, Prime Education, Revolutionizing Atopic Dermatitis, and Vindico Medical Education; and served on an advisory board for Merck. Melissa S. Seal, David Krupa, Diane Hanna, Scott Snyder, Patrick Burnett, David H. Chu, Robert C. Higham, and David R. Berk are employees of Arcutis Biotherapeutics. Erin Almaraz was an employee of Arcutis Biotherapeutics at the time of this research.
Figures
References
-
- Asher M. I., Montefort S., Björkstén B., et al., “Worldwide Time Trends in the Prevalence of Symptoms of Asthma, Allergic Rhinoconjunctivitis, and Eczema in Childhood: ISAAC Phases One and Three Repeat Multicountry Cross‐Sectional Surveys,” Lancet 368, no. 9537 (2006): 733–743. - PubMed
-
- Weidinger S., Simpson E. L., Silverberg J. I., et al., “Burden of Atopic Dermatitis in Paediatric Patients: An International Cross‐Sectional Study,” British Journal of Dermatology 190, no. 6 (2024): 846–857. - PubMed
-
- Lee E., Kim J. H., Cha H. R., et al., “Association Between Atopic Dermatitis and School Readiness in Preschool Children,” Pediatric Allergy and Immunology 34, no. 7 (2023): e13996. - PubMed
-
- Eucrisa (Crisaborole) Ointment 2% Prescribing Information (New York, NY: Pfizer Inc., 2023).
-
- Ahmed A., Solman L., and Williams H. C., “Magnitude of Benefit for Topical Crisaborole in the Treatment of Atopic Dermatitis in Children and Adults Does Not Look Promising: A Critical Appraisal,” British Journal of Dermatology 178, no. 3 (2018): 659–662. - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

