Role of tau versus TDP-43 pathology on medial temporal lobe atrophy in aging and Alzheimer's disease
- PMID: 39985478
- PMCID: PMC11846482
- DOI: 10.1002/alz.14582
Role of tau versus TDP-43 pathology on medial temporal lobe atrophy in aging and Alzheimer's disease
Abstract
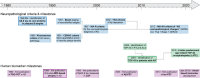
Hippocampal atrophy on magnetic resonance imaging is an important biomarker in Alzheimer's disease (AD). While hippocampal atrophy was thought to result from tau tangles in AD, different neuropathologies can lead to hippocampal atrophy, especially TAR DNA-binding protein 43 (TDP-43) pathology. In this narrative review, we evaluate existing studies on the relative contribution of tau and TDP-43 pathology to medial temporal lobe (MTL) atrophy. We report a clear association of both tau and TDP-43 neuropathology with MTL atrophy, even after correcting for other neuropathologies. Next, we discuss a potential synergism between tau and TDP-43 and the relative timing of the effects of both neuropathologies. Finally, avenues for future research will be discussed. A better understanding of the interplay between tau and TDP-43 neuropathologies and their effect on atrophy will help with the development of more specific biomarkers for limbic-predominant age-related TDP-43 encephalopathy and pinpointing of the optimal timing for testing anti-tau and anti-TDP-43 treatments in trials. HIGHLIGHTS: Both tau and TAR DNA-binding protein 43 (TDP-43) pathology contribute to medial temporal lobe atrophy. There is a positive association between tau and TDP-43 and potentially a synergism. It is unclear if tau and TDP-43 have an additive or synergistic effect on atrophy. The relative timing of the tau and TDP-43 effects on atrophy remains unclear. Clarifying the interplay between tau and TDP-43 will help improve magnetic resonance imaging biomarkers.
Keywords: Alzheimer's disease; TAR DNA‐binding protein 43; dementia; hippocampus; limbic‐predominant age‐related TDP‐43 encephalopathy; magnetic resonance imaging; medial temporal lobe; neurodegeneration; neurofibrillary tangle pathology.
© 2025 The Author(s). Alzheimer's & Dementia published by Wiley Periodicals LLC on behalf of Alzheimer's Association.
Conflict of interest statement
Unrelated to the content of this manuscript, Dr. Jagust has served as a consultant to Lilly and has equity in Molecular Medicine and Optoceutics. None of the other authors have anything to disclose.
Figures



Similar articles
-
Contribution of mixed pathology to medial temporal lobe atrophy in Alzheimer's disease.Alzheimers Dement. 2020 Jun;16(6):843-852. doi: 10.1002/alz.12079. Epub 2020 Apr 22. Alzheimers Dement. 2020. PMID: 32323446 Free PMC article.
-
Rates of hippocampal atrophy and presence of post-mortem TDP-43 in patients with Alzheimer's disease: a longitudinal retrospective study.Lancet Neurol. 2017 Nov;16(11):917-924. doi: 10.1016/S1474-4422(17)30284-3. Epub 2017 Sep 11. Lancet Neurol. 2017. PMID: 28919059 Free PMC article.
-
Downstream effects of polypathology on neurodegeneration of medial temporal lobe subregions.Acta Neuropathol Commun. 2021 Jul 21;9(1):128. doi: 10.1186/s40478-021-01225-3. Acta Neuropathol Commun. 2021. PMID: 34289895 Free PMC article.
-
Tau and TDP-43 synergy: a novel therapeutic target for sporadic late-onset Alzheimer's disease.Geroscience. 2021 Aug;43(4):1627-1634. doi: 10.1007/s11357-021-00407-0. Epub 2021 Jun 29. Geroscience. 2021. PMID: 34185246 Free PMC article. Review.
-
TDP-43 in aging and Alzheimer's disease - a review.Int J Clin Exp Pathol. 2011 Jan 30;4(2):147-55. Int J Clin Exp Pathol. 2011. PMID: 21326809 Free PMC article. Review.
Cited by
-
Alzheimer's disease and its co-pathologies: Implications for hippocampal degeneration, cognitive decline, and the role of APOE ε4.Alzheimers Dement. 2025 Jul;21(7):e70483. doi: 10.1002/alz.70483. Alzheimers Dement. 2025. PMID: 40665478 Free PMC article.
-
Tau, atrophy, and domain-specific cognitive impairment in typical Alzheimer's disease.Alzheimers Dement. 2025 Jul;21(7):e70511. doi: 10.1002/alz.70511. Alzheimers Dement. 2025. PMID: 40709494 Free PMC article.
-
Iron-Mediated Overexpression of Amyloid Precursor Protein via Iron Responsive mRNA in Alzheimer's Disease.Int J Mol Sci. 2025 May 30;26(11):5283. doi: 10.3390/ijms26115283. Int J Mol Sci. 2025. PMID: 40508094 Free PMC article. Review.
-
Looking into Abnormal Co-Expressions of Tau and TDP-43 in the Realm of Mixed Dementia Types: A Double-Punch Scenario.Brain Sci. 2025 Jul 3;15(7):716. doi: 10.3390/brainsci15070716. Brain Sci. 2025. PMID: 40722308 Free PMC article. Review.
-
The PREVENT-AD cohort: accelerating Alzheimer's disease research and treatment in Canada and beyond.medRxiv [Preprint]. 2025 Jul 23:2025.07.22.25331791. doi: 10.1101/2025.07.22.25331791. medRxiv. 2025. PMID: 40778177 Free PMC article. Preprint.
References
Publication types
MeSH terms
Substances
Grants and funding
- R01-AG070592/AG/NIA NIH HHS/United States
- 2022-00900/Vetenskapsrådet
- MultiPark
- AF-980872/Alzheimerfonden
- U19-AG069701/AG/NIA NIH HHS/United States
- R01 AG070592/AG/NIA NIH HHS/United States
- P30 AG062422/AG/NIA NIH HHS/United States
- AARG-22-926899/ALZ/Alzheimer's Association/United States
- 20210690/Crafoordska Stiftelsen
- R01 AG073282/AG/NIA NIH HHS/United States
- AF-993465/Alzheimerfonden
- U19 AG069701/AG/NIA NIH HHS/United States
- P30-AG062422/AG/NIA NIH HHS/United States
- R01 AG062542/AG/NIA NIH HHS/United States
- R01-AG073282/AG/NIA NIH HHS/United States
- R01-AG062542/AG/NIA NIH HHS/United States

