CD47 blockade reverses resistance to HDAC inhibitor by liberating anti-tumor capacity of macrophages
- PMID: 39994810
- PMCID: PMC11849317
- DOI: 10.1186/s13046-025-03335-5
CD47 blockade reverses resistance to HDAC inhibitor by liberating anti-tumor capacity of macrophages
Abstract
Background: Targeting oncogenic histone modification by histone deacetylase inhibitors (HDACis) demonstrates promising prospects in clinical cancer treatment, whereas a notable proportion of patients cannot benefit from HDACi therapy. This study aims to explore how HDACi influences the tumor microenvironment, in order to identify potential targets for reversing the resistance to HDACi therapies.
Methods: Macrophage infiltration was compared between HDACi-responding and HDACi-nonresponding cancer patients. The impact of HDACis on the phagocytic capacity of macrophages was investigated through macrophage-tumor cell co-culture system. CD47 expression in tumor cell lines and patient-derived organoids was evaluated by quantitative polymerase chain reaction (QPCR) and flow cytometry. Mechanistic studies were conducted through co-immunoprecipitation (co-IP) and chromatin immunoprecipitation (ChIP). The synergistic effect of HDACis and CD47 neutralizing antibody was assessed in subcutaneous murine tumor models. Bioinformatics approaches were adopted to analyze how macrophage infiltration determines the prognostic significance of CD47 expression in cancer patients.
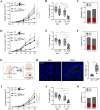
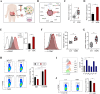
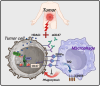
Results: High macrophage infiltration is a determinant of therapeutic non-response to HDACi, cancer patients who did not respond to HDACi exhibit massive infiltration of tumor-associated macrophages (TAMs). TAM depletion reversed the resistance to HDACi therapy. Mechanistically, HDACi impaired the phagocytic capacity of macrophages against tumor cells through epigenetically upregulating CD47 expression. Reciprocally, HDACi-upregulated CD47 polarized macrophages towards a pro-tumor M2 phenotype through SIRPα ligation. In tumor-bearing mice, HDACi monotherapy only marginally delayed tumor progression, while the concurrent neutralization of CD47 exhibited potent anti-tumor effect through re-educating TAMs towards a tumoricidal phenotype. In cancer patients, CD47 was found to determine the prognostic significance of TAMs.
Conclusions: Our study offers a rationale for targeting macrophage infiltration or blocking CD47 to sensitize HDACi therapies in cancer patients.
Keywords: CD47; Cancer; HDAC inhibitor; Macrophage; Tumor microenvironment.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Ethics approval and consent to participate: Experiments using clinical specimens were conducted under the approval from the Medical Ethics Committee of Sir Run Run Shaw Hospital of Zhejiang University (20220103–56), and the First Affiliated Hospital of Huzhou University (2021KYLL-Y-005). Informed consents were obtained from all patients. Consent for publication: Not applicable. Competing interests: The authors declare no competing interests.
Figures





References
-
- Dawson MA, Kouzarides T. Cancer epigenetics: from mechanism to therapy. Cell. 2012;150(1):12–27. - PubMed
-
- Cheng B, Pan W, Xiao Y, Ding Z, Zhou Y, Fei X, et al. HDAC-targeting epigenetic modulators for cancer immunotherapy. Eur J Med Chem. 2024;265:116129. - PubMed
-
- Gilbert J, Gore SD, Herman JG, Carducci MA. The clinical application of targeting cancer through histone acetylation and hypomethylation. Clin Cancer Res. 2004;10(14):4589–96. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

