Capnometry-Guided Respiratory Intervention in Veteran PTSD: Impact on Symptom Clusters
- PMID: 39997265
- PMCID: PMC11855826
- DOI: 10.3390/healthcare13040390
Capnometry-Guided Respiratory Intervention in Veteran PTSD: Impact on Symptom Clusters
Abstract
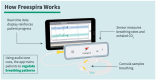
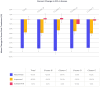
Background/Objectives: Post-traumatic stress disorder (PTSD) is a challenging psychiatric condition to treat, with suboptimal recovery and difficulty tolerating exposure-based psychotherapies often noted in outcomes research. The aim of this study was to examine patterns of symptom reduction in veterans with PTSD treated with a Capnometry-Guided Respiratory Intervention (CGRI), a 28-day treatment teaching about the normalization of respiratory rate and exhaled carbon dioxide levels via biofeedback. We hypothesized reductions in total PCL-5 scores and all symptom clusters immediately post-treatment but with relative resistance to changes in hyperarousal symptoms, as reported in the outcomes of research using other evidence-based psychotherapies. Methods: In this report of real-world outcomes, we included 164 veterans treated with CGRI. Pre- and post-treatment PTSD Checklists for DSM-5 (PCL-5) scales were recorded and analyzed based on the total, cluster, and item scores. Subjects were additionally classified into Recovered, Improved, or Suboptimal subgroups based on their response to treatment. Data were compiled during routine clinical care and are available for retrospective analysis. Results: Treatment response was reported in 53% of participants, with a mean total PCL-5 score reduction of 12 points post-treatment (effect size, Glass's Δ = 0.99, large) and individual PCL-5 clusters showing medium to large effect sizes (effect size = 0.71 to 0.98). Contrary to our hypothesis, a large effect size was found in the hyperarousal cluster, with post-treatment scores being significantly improved compared to pre-treatment scores (effect size = 0.98). In the Recovered group, all 20 PCL-5 items showed significant declines, while significant reductions were reported in some items in the Improved group and no item improvements were noted in the Suboptimal group. Conclusions: Consistently with prior published trials reporting overall improvements in PTSD symptoms, in this report, the CGRI produced clinically meaningful reductions in PCL-5 cluster scores in addition to total scores. Unlike reports from several trials of cognitive therapies, this study found hyperarousal symptoms to be responsive to treatment. The CGRI shows evidence of improvement across the range of PTSD symptoms in the immediate post-treatment interval. The absence of an extended post-treatment follow up introduces uncertainty concerning the durability of benefits experienced, although previous CGRI research on both panic disorder and PTSD has shown the maintenance of symptom reduction in six- to twelve-month intervals.
Keywords: CGRI; Freespira; PTSD; biofeedback; carbon dioxide sensitivity; digital therapeutic; hyperarousal; panic disorder; stress disorders; trauma.
Conflict of interest statement
Robert N. Cuyler is employed as Chief Clinical Officer at Freespira, Inc. and holds stock options in the company. Juliette S. Mojgani does not have conflicts of interest to declare. Julio Cezar Albuquerque da Costa does not have conflicts of interest to declare. Rafael Freire is the primary investigator in a grant-funded clinical trial sponsored by Biohaven Pharmaceuticals. The authors declare that this study received funding from Freespira, Inc. The funder was not involved in the study design, collection, analysis, interpretation of data, the writing of this article or the decision to submit it for publication.
Figures
Similar articles
-
Real-world outcomes of an innovative digital therapeutic for treatment of panic disorder and PTSD: A 1,500 patient effectiveness study.Front Digit Health. 2022 Nov 17;4:976001. doi: 10.3389/fdgth.2022.976001. eCollection 2022. Front Digit Health. 2022. PMID: 36465089 Free PMC article.
-
Investigation of a Capnometry Guided Respiratory Intervention in the Treatment of Posttraumatic Stress Disorder.Appl Psychophysiol Biofeedback. 2021 Dec;46(4):367-376. doi: 10.1007/s10484-021-09521-3. Appl Psychophysiol Biofeedback. 2021. PMID: 34468913 Free PMC article.
-
Trauma-Sensitive Yoga for Post-Traumatic Stress Disorder in Women Veterans who Experienced Military Sexual Trauma: Interim Results from a Randomized Controlled Trial.J Altern Complement Med. 2021 Mar;27(S1):S45-S59. doi: 10.1089/acm.2020.0417. J Altern Complement Med. 2021. PMID: 33788599 Clinical Trial.
-
Psychotherapy for Military-Related PTSD: A Review of Randomized Clinical Trials.JAMA. 2015 Aug 4;314(5):489-500. doi: 10.1001/jama.2015.8370. JAMA. 2015. PMID: 26241600 Review.
-
Psychological therapies for the treatment of post-traumatic stress disorder in children and adolescents (Review).Evid Based Child Health. 2013 May;8(3):1004-116. doi: 10.1002/ebch.1916. Evid Based Child Health. 2013. PMID: 23877914 Review.
References
-
- Schein J., Houle C., Urganus A., Cloutier M., Patterson-Lomba O., Wang Y., King S., Levinson W., Guérin A., Lefebvre P., et al. Prevalence of post-traumatic stress disorder in the United States: A systematic literature review. Curr. Med. Res. Opin. 2021;37:2151–2161. doi: 10.1080/03007995.2021.1978417. - DOI - PubMed
-
- Pietrzak R.H., Goldstein R.B., Southwick S.M., Grant B.F. Medical comorbidity of full and partial posttraumatic stress disorder in US adults: Results from Wave 2 of the National Epidemiologic Survey on Alcohol and Related Conditions. Psychosom. Med. 2011;73:697–707. doi: 10.1097/PSY.0b013e3182303775. - DOI - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources