Bone marrow mesenchymal stem cells alleviate neurological dysfunction by reducing autophagy damage via downregulation of SYNPO2 in neonatal hypoxic-ischemic encephalopathy rats
- PMID: 40000609
- PMCID: PMC11862179
- DOI: 10.1038/s41419-025-07439-w
Bone marrow mesenchymal stem cells alleviate neurological dysfunction by reducing autophagy damage via downregulation of SYNPO2 in neonatal hypoxic-ischemic encephalopathy rats
Abstract
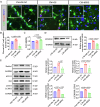
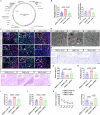
Neonatal hypoxic-ischemic encephalopathy (HIE) is worsened by autophagy-induced neuronal damage, with SYNPO2 playing a key role in this process. This study investigates the involvement of SYNPO2 in neuronal autophagy and explores the potential of bone marrow mesenchymal stem cells (BMSCs) to alleviate HIE-induced dysfunction by inhibiting SYNPO2-mediated autophagy. Using in vitro and in vivo neonatal HIE models, we observed an upregulation of SYNPO2 expression, accompanied by increased neuronal injury and aggregation of autophagy-related proteins. Intervention with BMSCs effectively reduced SYNPO2 expression, and SYNPO2 depression mitigated neuroautophagic damage and improved neurological dysfunctions. Moreover, SYNPO2 overexpression exacerbated neuroautophagy despite BMSC treatment, while SYNPO2 depletion notably reduced neuroautophagic damage and alleviated cognitive impairments, retaining the neuroprotective efficacy of BMSC treatment. These findings confirm the role of BMSCs in attenuating HIE injury by suppressing neuroautophagy and provide insights into the mechanistic involvement of SYNPO2. Ultimately, this study identifies SYNPO2 as a novel therapeutic target for neonatal HIE and supports the clinical potential of BMSCs in HIE management.
© 2025. The Author(s).
Conflict of interest statement
Competing interests: The authors declare no competing interests. Ethics approval: All animal experiments were approved by the Experimental Animal Ethics Committee of Zunyi Medical University ([2020]2-097), and conducted in compliance with Guide for the Care and Use of Laboratory Animals published by the National Institutes of Health.
Figures




References
-
- McIntyre S, Nelson KB, Mulkey SB, Lechpammer M, Molloy E, Badawi N. Neonatal encephalopathy: Focus on epidemiology and underexplored aspects of etiology. Semin Fetal Neonatal Med. 2021;26:101265. - PubMed
-
- Yang L, Zhao H, Cui H. Treatment and new progress of neonatal hypoxic-ischemic brain damage. Histol Histopathol. 2020;35:929–36. - PubMed
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

