Immunohistochemical Analysis of Inter-Alpha-Trypsin Inhibitor Heavy Chain 2 and Enolase 1 in Canine Mammary Tumors: Associations with Tumor Aggressiveness and Prognostic Significance
- PMID: 40005870
- PMCID: PMC11860171
- DOI: 10.3390/vetsci12020110
Immunohistochemical Analysis of Inter-Alpha-Trypsin Inhibitor Heavy Chain 2 and Enolase 1 in Canine Mammary Tumors: Associations with Tumor Aggressiveness and Prognostic Significance
Abstract
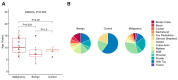
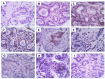
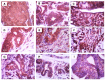
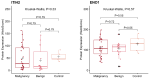
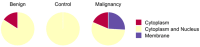
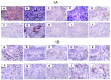
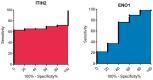
Mammary neoplasms in dogs are a common clinical concern, especially in middle-aged and older intact females. These tumors share similarities with human breast cancer in terms of histology, disease progression, and risk factors, making dogs a relevant model for breast cancer research. The search for biomarkers in canine mammary tumors is essential to understand tumor progression and identify potential therapeutic targets. This study investigated the expression of two potential biomarkers-Inter-Alpha-Trypsin Inhibitor Heavy Chain 2 (ITIH2) and Enolase 1 (ENO1)-in the mammary glands of healthy and tumor-bearing dogs using immunohistochemistry. Both proteins were identified in previous proteomic analyses of extracellular vesicles derived from the plasma of healthy and tumor-bearing dogs. A total of fifty-one canine mammary tissue samples were analyzed and categorized into three groups: (i) the control group, composed of five samples of normal mammary tissue without neoplasia; (ii) benign tumors, composed of nineteen samples of benign mixed tumors; and (iii) malignant tumors, which included six carcinomas in grade 1 mixed tumors, five carcinomas in grade 2 mixed tumors, thirteen solid carcinomas of grade 3, one papillary carcinoma, and two tubular carcinomas. Regarding the intensity of staining, quantified by histoscore, there were no significant differences in the comparison between the groups; for ITIH2, the p-value was 0.33, and for ENO1, the p-value was 0.57. Regarding the predictive potential of their respective ROC curves, the proteins demonstrated low predictive power in canine mammary tumors. These findings indicate that neither ITIH2 nor ENO1 demonstrated strong prognostic value in this setting, as demonstrated by their moderate AUC values, wide confidence intervals, and lack of statistical significance. However, this study found distinct tissue localization patterns for ITIH2 and subcellular localization for ENO1. As an additional way to examine possible associations of these proteins with epithelial-mesenchymal transition, the ZEB1 antibody was tested by both single and double immunohistochemistry, demonstrating a tendency to be more intensely expressed in the malignant group and tending to be associated with ENO1 in canine mammary tumors.
Keywords: biomarkers; bitches; extracellular vesicles (EVs); mammary tumors; prognosis.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures


Similar articles
-
Overexpression of α-enolase correlates with poor survival in canine mammary carcinoma.BMC Vet Res. 2011 Oct 21;7:62. doi: 10.1186/1746-6148-7-62. BMC Vet Res. 2011. PMID: 22014164 Free PMC article.
-
Exploring Canine Mammary Cancer through Liquid Biopsy: Proteomic Profiling of Small Extracellular Vesicles.Cancers (Basel). 2024 Jul 17;16(14):2562. doi: 10.3390/cancers16142562. Cancers (Basel). 2024. PMID: 39061201 Free PMC article.
-
Diagnosis, classification and grading of canine mammary tumours as a model to study human breast cancer: an Clinico-Cytohistopathological study with environmental factors influencing public health and medicine.Cancer Cell Int. 2013 Aug 9;13:79. doi: 10.1186/1475-2867-13-79. eCollection 2013. Cancer Cell Int. 2013. Retraction in: Cancer Cell Int. 2016 Nov 4;16:83. doi: 10.1186/s12935-016-0358-6. PMID: 23937693 Free PMC article. Retracted.
-
A comparative study of canine and human breast cancer.Invest Cell Pathol. 1979 Oct-Dec;2(4):257-75. Invest Cell Pathol. 1979. PMID: 396282 Review.
-
Canine, Feline, and Murine Mammary Tumors as a Model for Translational Research in Breast Cancer.Vet Sci. 2025 Feb 19;12(2):189. doi: 10.3390/vetsci12020189. Vet Sci. 2025. PMID: 40005948 Free PMC article. Review.
References
-
- Sorenmo K.U., Kristiansen V.M., Cofone M.A., Shofer F.S., Breen A.M., Langeland M., Mongil C.M., Grondahl A.M., Teige J., Goldschmidt M.H. Canine mammary gland tumours, a histological continuum from benign to malignant; clinical and histopathological evidence. Veter- Comp. Oncol. 2009;7:162–172. doi: 10.1111/j.1476-5829.2009.00184.x. - DOI - PubMed
-
- Zuccari D.A.P.D.C., Santana A.E., Rocha N.S. Correlação entre a citologia aspirativa por agulha fina e a histologia no diagnóstico de tumores mamários de cadelas. Braz. J. Vet. Res. Anim. Sci. 2001;38:38–41. doi: 10.1590/S1413-95962001000100007. - DOI
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

