Alternaria alternata (Fr) Keissl Crude Extract Inhibits HIV Subtypes and Integrase Drug-Resistant Strains at Different Stages of HIV Replication
- PMID: 40006004
- PMCID: PMC11859181
- DOI: 10.3390/ph18020189
Alternaria alternata (Fr) Keissl Crude Extract Inhibits HIV Subtypes and Integrase Drug-Resistant Strains at Different Stages of HIV Replication
Abstract
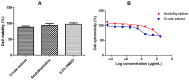
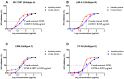
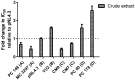
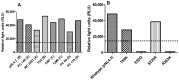
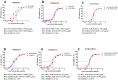
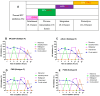
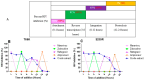
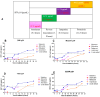
Background/Objectives: The development of HIV drug resistance to current antiretrovirals, and the antiretrovirals' inability to cure HIV, provides the need of developing novel drugs that inhibit HIV-1 subtypes and drug-resistance strains. Fungal endophytes, including Alternaria alternata, stand out for their potentially antiviral secondary metabolites. Hence, this study investigates the anti-HIV activities and mechanism of action of the A. alternata crude extract against different HIV-1 subtypes and integrase-resistant mutant strains. Methods: Cytotoxicity of the A. alternata crude extract on TZM-bl cells using the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide (MTT) assay was performed. The crude extract antiviral activity against subtypes A, B, C, and D and integrase drug-resistant strain T66K and S230R was determined using a luciferase-based antiviral assay. Luciferase and p24 ELISA-based time-of-addition assays were used to determine the mechanism of action of the crude extract. Docking scores and protein ligand interactions of integrase T66K and S230R strains against the identified bioactive compounds were determined. Results: The crude extract CC50 was 300 μg/mL and not cytotoxic to the TZM-bl cell lines. In HIV-1 subtypes A, B, C, and D, the crude extract exhibited 100% inhibition and therapeutic potential. The A. alternata crude extract had strong anti-HIV-1 activity against integrase strand transfer drug-resistant strains T66K and S230R, with a 0.7265- and 0. 8751-fold increase in susceptibility. The crude extract had antiviral activity during attachment, reverse transcription, integration, and proteolysis. In silico calculations showed compounds 2,3-2H-Benzofuran-2-one, 3,3,4,6-tetramethyl-, 3-Methyl-1,4-diazabicyclo[4.3.0]nonan-2,5-dione, N-acetyl, Coumarin, 3,4-dihydro-4,5,7-trimethyl-, Cyclopropanecarboxamide, N-cycloheptyl, Pyrrolo[1,2-a]pyrazine-1,4-dione, and hexahydro-3-(2-methylpropyl)- crude extract bioactive compounds had strong docking scores and diverse binding mechanisms with integrase. Conclusions: The A. alternata crude extract demonstrates strong antiviral activity against different HIV-1 subtypes and integrase drug-resistance strains. The extract inhibited various stages of the HIV-1 life cycle. The bioactive compounds 2,3-2H-Benzofuran-2-one, 3,3,4,6-tetramethyl-, 3-Methyl-1,4-diazabicyclo[4.3.0]nonan-2,5-dione, N-acetyl, Coumarin, 3,4-dihydro-4,5,7-trimethyl-, Cyclopropanecarboxamide, N-cycloheptyl, Pyrrolo[1,2-a]pyrazine-1,4-dione, and hexahydro-3-(2-methylpropyl)- may be responsible for the antiviral activity of A. alternata.
Keywords: A. alternata; HIV-subtypes; anti-HIV activity; integrase inhibitors; molecular docking.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures

Similar articles
-
Anti-HIV Activity and Immunomodulatory Properties of Fractionated Crude Extracts of Alternaria alternata.Microorganisms. 2024 Jun 5;12(6):1150. doi: 10.3390/microorganisms12061150. Microorganisms. 2024. PMID: 38930532 Free PMC article.
-
Secondary metabolites produced by endophytic fungi, Alternaria alternata, as potential inhibitors of the human immunodeficiency virus.Front Genet. 2022 Dec 13;13:1077159. doi: 10.3389/fgene.2022.1077159. eCollection 2022. Front Genet. 2022. PMID: 36583026 Free PMC article.
-
Study on suitable analysis method for HIV-1 non-catalytic integrase inhibitor.Virol J. 2021 Jan 13;18(1):17. doi: 10.1186/s12985-020-01476-x. Virol J. 2021. PMID: 33436020 Free PMC article.
-
A computational overview of integrase strand transfer inhibitors (INSTIs) against emerging and evolving drug-resistant HIV-1 integrase mutants.Arch Microbiol. 2023 Mar 26;205(4):142. doi: 10.1007/s00203-023-03461-8. Arch Microbiol. 2023. PMID: 36966200 Free PMC article. Review.
-
Characterization and structural analysis of HIV-1 integrase conservation.AIDS Rev. 2009 Jan-Mar;11(1):17-29. AIDS Rev. 2009. PMID: 19290031 Review.
Cited by
-
Synthesis, Characterisation, Biological Evaluation and In Silico Studies of Quinoline-1,2,3-Triazole-Anilines as Potential Antitubercular and Anti-HIV Agents.Molecules. 2025 May 10;30(10):2119. doi: 10.3390/molecules30102119. Molecules. 2025. PMID: 40430292 Free PMC article.
References
-
- UNAIDS R. Global AIDS report. Geneva Switzerland. 2024. [(accessed on 21 August 2024)]. Available online: www.unaids.org/en/resources/documents/2024/global-aids-update-2024.
Grants and funding
LinkOut - more resources
Full Text Sources

