Pd-Catalyzed Strain-Releasing Dyotropic Rearrangement: Ring-Expanding Amidofluorination of Methylenecyclobutanes
- PMID: 40023786
- PMCID: PMC11925333
- DOI: 10.1021/jacs.5c01108
Pd-Catalyzed Strain-Releasing Dyotropic Rearrangement: Ring-Expanding Amidofluorination of Methylenecyclobutanes
Abstract
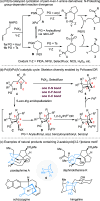
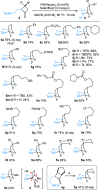
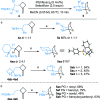
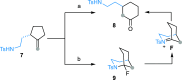
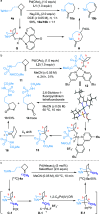
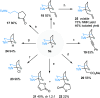
Under the Pd(II)/Pd(IV) catalytic cycle, the cyclization of pent-4-en-1-amine derivatives typically yields either pyrrolidines or piperidines depending on the N-protecting group. We report herein an unprecedented Pd(II)-catalyzed oxidative domino process that converts readily accessible N-protected 2-(2-amidoethyl)-1-methylenecyclobutane derivatives to 1-fluoro-2-azabicyclo[3.2.1]octanes. This transformation constructs three chemical bonds under mild conditions [Pd(hfacac)2 (5.0 mol %), Selectfluor (2.0 equiv), MeCN, 60 °C, 10 min] through a domino sequence involving 5-exo-trig amidopalladation/Pd(II)-oxidation/chemoselective dyotropic rearrangement/C-F bond-forming reductive elimination. Notably, the cyclization mode remains independent of the N-protecting group under these conditions. Furthermore, diverse functional groups can be introduced at the bridgehead position of a bicyclic compound via an apparent anti-Bredt bridgehead iminium intermediate.
Conflict of interest statement
The authors declare no competing financial interest.
Figures
Similar articles
-
Diverting the 5-exo-Trig Oxypalladation to Formally 6-endo-Trig Fluorocycloetherification Product through 1,2-O/Pd(IV) Dyotropic Rearrangement.J Am Chem Soc. 2023 Jul 26;145(29):15735-15741. doi: 10.1021/jacs.3c06158. Epub 2023 Jul 18. J Am Chem Soc. 2023. PMID: 37462356
-
Apparent 6-endo-trig Carbofluorination of Alkenes Enabled by Palladium-Based Dyotropic Rearrangement.Angew Chem Int Ed Engl. 2022 Dec 12;61(50):e202211470. doi: 10.1002/anie.202211470. Epub 2022 Nov 16. Angew Chem Int Ed Engl. 2022. PMID: 36222430
-
Chemoselectivity in Pd-Based Dyotropic Rearrangement: Development and Application in Total Synthesis of Pheromones.J Am Chem Soc. 2025 Jan 15;147(2):2077-2085. doi: 10.1021/jacs.4c15764. Epub 2024 Dec 30. J Am Chem Soc. 2025. PMID: 39812086
-
Chemoselective Pd-Based Dyotropic Rearrangement: Fluorocyclization and Regioselective Wacker Reaction of Homoallylic Amides.J Am Chem Soc. 2024 Nov 6;146(44):30014-30019. doi: 10.1021/jacs.4c13359. Epub 2024 Oct 25. J Am Chem Soc. 2024. PMID: 39453186
-
The organometallic fluorine chemistry of palladium and rhodium: studies toward aromatic fluorination.Acc Chem Res. 2010 Jan 19;43(1):160-71. doi: 10.1021/ar9001763. Acc Chem Res. 2010. PMID: 19788304 Review.
References
-
-
Pd(0)-catalyzed diamination of 1,3-dienes, see:
- Du H.; Zhao B.; Shi Y. A Facile Pd(0)-Catalyzed Regio- and Stereoselective Diamination of Conjugated Dienes and Trienes. J. Am. Chem. Soc. 2007, 129, 762–763. 10.1021/ja0680562. - DOI - PubMed
- Du H.; Yuan W.; Zhao B.; Shi Y. Catalytic Asymmetric Diamination of Conjugated Dienes and Triene. J. Am. Chem. Soc. 2007, 129, 11688–11689. 10.1021/ja074698t. - DOI - PubMed
- Zhao B.; Du H.; Cui S.; Shi Y. Synthetic and Mechanistic Studies on Pd(0)-Catalyzed Diamination of Conjugated Dienes. J. Am. Chem. Soc. 2010, 132, 3523–3532. 10.1021/ja909459h. - DOI - PMC - PubMed
- Zhu Y.; Cornwall R. G.; Du H.; Zhao B.; Shi Y. Catalytic Diamination of Olefins via N–N Bond Activation. Acc. Chem. Res. 2014, 47, 3665–3678. 10.1021/ar500344t. - DOI - PMC - PubMed
-
-
-
Pd(II) catalyzed diamination of 1,3-dienes, see:
- Bar G. L. J.; Lloyd-Jones G. C.; Booker-Milburn K. I. Pd(II)-Catalyzed Intermolecular 1,2-Diamination of Conjugated Dienes. J. Am. Chem. Soc. 2005, 127, 7308–7309. 10.1021/ja051181d. - DOI - PubMed
- Wu Z.; Wen K.; Zhang J.; Zhang W. Pd(II)-Catalyzed Aerobic Intermolecular 1,2-Diamination of Conjugated Dienes: A Regio- and Chemoselective [4 + 2] Annulation for the Synthesis of Tetrahydroquinoxalines. Org. Lett. 2017, 19, 2813–2816. 10.1021/acs.orglett.7b00919. - DOI - PubMed
-
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

