Disruption of the moonlighting function of CTF18 in a patient with T-lymphopenia
- PMID: 40028343
- PMCID: PMC11868726
- DOI: 10.3389/fimmu.2025.1539848
Disruption of the moonlighting function of CTF18 in a patient with T-lymphopenia
Abstract
Introduction: Newborn screening for immunodeficiency has led to the identification of numerous cases for which the causal etiology is unknown.
Methods: Here we report the diagnosis of T lymphopenia of unknown etiology in a male proband. Whole exome sequencing (WES) was employed to nominate candidate variants, which were then analyzed functionally in zebrafish and in mice bearing orthologous mutations.
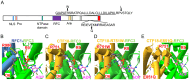
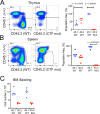
Results: WES revealed missense mutations in CHTF18 that were inherited in an autosomal recessive manner. CTF18, encoded by the CHTF18 gene, is a component of a secondary clamp loader, which is primarily thought to function by promoting DNA replication. We determined that the patient's variants in CHTF18 (CTF18 R751W and E851Q) were damaging to function and severely attenuated the capacity of CTF18 to support hematopoiesis and lymphoid development, strongly suggesting that they were responsible for his T lymphopenia; however, the function of CTF18 appeared to be unrelated to its role as a clamp loader. DNA-damage, expected when replication is impaired, was not evident by expression profiling in murine Chtf18 mutant hematopoietic stem and progenitor cells (HSPC), nor was development of Ctf18-deficient progenitors rescued by p53 loss. Instead, we observed an expression signature suggesting disruption of HSPC positioning and migration. Indeed, the positioning of HSPC in ctf18 morphant zebrafish embryos was perturbed, suggesting that HSPC function was impaired through disrupted positioning in hematopoietic organs.
Discussion: Accordingly, we propose that T lymphopenia in our patient resulted from disturbed cell-cell contacts and migration of HSPC, caused by a non-canonical function of CHTF18 in regulating gene expression.
Keywords: CHTF18; T lymphocyte; immunodeficiency; thymus; zebrafish.
Copyright © 2025 Sertori, Truong, Singh, Shinton, Price, Sharo, Shultes, Sunderam, Rana, Srinivasan, Datta, Font-Burgada, Brenner, Puck and Wiest.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest. The author(s) declared that they were an editorial board member of Frontiers, at the time of submission. This had no impact on the peer review process and the final decision.
Figures





Similar articles
-
Investigation of the causal etiology in a patient with T-B+NK+ immunodeficiency.Front Immunol. 2022 Jul 29;13:928252. doi: 10.3389/fimmu.2022.928252. eCollection 2022. Front Immunol. 2022. PMID: 35967429 Free PMC article.
-
Exome Sequencing Diagnoses X-Linked Moesin-Associated Immunodeficiency in a Primary Immunodeficiency Case.Front Immunol. 2018 Mar 5;9:420. doi: 10.3389/fimmu.2018.00420. eCollection 2018. Front Immunol. 2018. PMID: 29556235 Free PMC article.
-
Disruption of CHTF18 causes defective meiotic recombination in male mice.PLoS Genet. 2012;8(11):e1002996. doi: 10.1371/journal.pgen.1002996. Epub 2012 Nov 1. PLoS Genet. 2012. PMID: 23133398 Free PMC article.
-
When Screening for Severe Combined Immunodeficiency (SCID) with T Cell Receptor Excision Circles Is Not SCID: a Case-Based Review.J Clin Immunol. 2021 Feb;41(2):294-302. doi: 10.1007/s10875-020-00931-2. Epub 2021 Jan 7. J Clin Immunol. 2021. PMID: 33411155 Free PMC article. Review.
-
Genetically determined lymphopenia and autoimmune manifestations.Curr Opin Immunol. 2008 Jun;20(3):318-24. doi: 10.1016/j.coi.2008.02.001. Epub 2008 Apr 9. Curr Opin Immunol. 2008. PMID: 18403192 Review.
References
-
- Dvorak CC, Haddad E, Heimall J, Dunn E, Buckley RH, Kohn DB, et al. . The diagnosis of severe combined immunodeficiency (SCID): The Primary Immune Deficiency Treatment Consortium (PIDTC) 2022 Definitions. J Allergy Clin Immunol. (2023) 151:539–46. doi: 10.1016/j.jaci.2022.1010.1022 - DOI - PMC - PubMed
-
- Thakar MS, Logan BR, Puck JM, Dunn EA, Buckley RH, Cowan MJ, et al. . Measuring the effect of newborn screening on survival after haematopoietic cell transplantation for severe combined immunodeficiency: a 36-year longitudinal study from the Primary Immune Deficiency Treatment Consortium. Lancet. (2023) 402:129–40. doi: 10.1016/S0140-6736(1023)00731-00736 - DOI - PMC - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

