Liquid biopsy for assessment of RFS (recurrence-free survival) in NSCLC (non-small cell lung cancer) patients post-treatment through circulating tumour DNA detection: A meta-analysis
- PMID: 40028487
- PMCID: PMC11863698
- DOI: 10.1016/j.jlb.2023.100127
Liquid biopsy for assessment of RFS (recurrence-free survival) in NSCLC (non-small cell lung cancer) patients post-treatment through circulating tumour DNA detection: A meta-analysis
Abstract
Background: One of the main techniques for diagnosing lung cancer is still tissue biopsy. The more recent liquid biopsy technique includes the assessment of fragments of cfDNA, ctDNA, and CTCs, which are released by tumour cells and act as biomarkers. After compiling information from the existing literature, we conducted a statistical analysis to evaluate the prognosis and recurrence of NSCLC post-treatment using ctDNA levels.
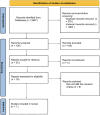
Method: Based on predefined inclusion and exclusion criteria, published studies between 2000 and 2022 from electronic databases with statistical data on ctDNA levels for NSCLC were included. Then, using PRISMA guidelines, we carried out a systematic review of those studies.
Result: A total of 7 studies were included in the analysis, and we found a correlation of ctDNA levels for determination of prognosis and recurrence after treatment in NSCLC. Meta-analysis was performed using RevMan version 5.4. A P-value of less than 0.05 was considered significant. Studies were divided into three groups: <1 week post-surgery, one week to 3 months post-surgery, and > three months post-surgery. Analysis showed that in <1-week post-surgery group, ctDNA-negative post-treatment detection had an HR of 4.85 (95 % CI: 2.41 to 9.77; p=<0.0001, I2 = 0 %) for recurrence-free survival compared to ctDNA-positive post-treatment detection. Similar findings were obtained for one week to 3 months and > three months group, with HR of 5.27 (95 % CI: 3.62 to 7.67; p=<0.00001, I2 = 11 %) and HR of 6.62 (95 % CI: 2.85 to 15.35; p=<0.0001, I2 = 64 %) respectively.
Conclusion: According to our meta-analysis, patients with NSCLC who were ctDNA negative after surgery had a significantly longer recurrence-free survival than those who were ctDNA positive. As a result, liquid biopsy used to measure ctDNA levels is a useful non-invasive technique to assess the prognosis in such patients. The early detection of recurrence may also allow timely therapeutic intervention, leading to better outcomes.
Keywords: Liquid biopsy; Minimal residual disease (MRD); Non-small cell lung cancer prognosis after treatment; Recurrence-free survival in non-small cell lung cancer patients; ctDNA; ctDNA detection.
© 2023 The Authors.
Conflict of interest statement
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures


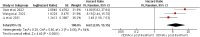
Similar articles
-
The role of liquid biopsy in predicting post-operative recurrence of non-small cell lung cancer.J Thorac Dis. 2018 Apr;10(Suppl 7):S838-S845. doi: 10.21037/jtd.2018.04.08. J Thorac Dis. 2018. PMID: 29780630 Free PMC article.
-
Perioperative circulating tumor DNA as a potential prognostic marker for operable stage I to IIIA non-small cell lung cancer.Cancer. 2022 Feb 15;128(4):708-718. doi: 10.1002/cncr.33985. Epub 2021 Oct 18. Cancer. 2022. PMID: 35076939
-
Potential clinical utility of liquid biopsy in early-stage non-small cell lung cancer.BMC Med. 2022 Dec 14;20(1):480. doi: 10.1186/s12916-022-02681-x. BMC Med. 2022. PMID: 36514063 Free PMC article.
-
Association of Circulating Tumor DNA With Disease-Free Survival in Breast Cancer: A Systematic Review and Meta-analysis.JAMA Netw Open. 2020 Nov 2;3(11):e2026921. doi: 10.1001/jamanetworkopen.2020.26921. JAMA Netw Open. 2020. PMID: 33211112 Free PMC article.
-
Circulating tumour DNA-Based molecular residual disease detection in resectable cancers: a systematic review and meta-analysis.EBioMedicine. 2024 May;103:105109. doi: 10.1016/j.ebiom.2024.105109. Epub 2024 Apr 13. EBioMedicine. 2024. PMID: 38614009 Free PMC article.
References
Publication types
LinkOut - more resources
Full Text Sources
Research Materials

