Induction Camrelizumab and Modified TPF (Nab-Paclitaxel, Cisplatin, and S-1) in Locoregionally Advanced Nasopharyngeal Carcinoma
- PMID: 40031812
- PMCID: PMC12127089
- DOI: 10.1111/cas.70037
Induction Camrelizumab and Modified TPF (Nab-Paclitaxel, Cisplatin, and S-1) in Locoregionally Advanced Nasopharyngeal Carcinoma
Abstract
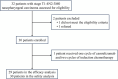
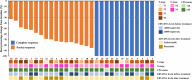
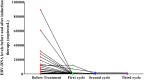
Induction chemo-immunotherapy has emerged as a potential treatment option for locoregionally advanced nasopharyngeal carcinoma (LANPC). This study aimed to evaluate the efficacy and safety of camrelizumab combined with modified TPF (nab-paclitaxel, cisplatin, and S-1) as induction chemo-immunotherapy in LANPC. Patients with stage T1-4N2-3M0 NPC who received induction chemo-immunotherapy were enrolled from July 2023 to May 2024. They underwent three cycles of chemo-immunotherapy, including camrelizumab 200 mg on day 1, nab-paclitaxel 260 mg/m2 on day 1, cisplatin 25 mg/m2 on days 1-3, and oral S-1 40-60 mg twice daily from days 1 to 14, every 21 days. The primary endpoint was the complete response (CR) rate, while secondary endpoints included the safety and objective response rate (ORR). A total of 30 patients were enrolled, with 29 (96.7%) completing three cycles of induction chemo-immunotherapy. The CR rate was 41.4% (12/29), achieving the predefined endpoint. The CR rate for the primary nasopharyngeal tumor and cervical lymph nodes was both 65.5% (19/29). Seventeen patients achieved a partial response (PR), resulting in an ORR of 100%. Grade 3 or 4 chemotherapy-related adverse events occurred in 26.6% of patients. Immune-related adverse events of any grade were reported in 20 (66.7%) patients, including reactive cutaneous capillary endothelial proliferation in 10 patients (40.0%), all of which were Grade 1 or 2. One patient (3.5%) experienced a Grade 3 rash. No treatment-related deaths occurred. Our study suggests that induction chemo-immunotherapy of camrelizumab plus modified TPF demonstrated an excellent CR rate and an acceptable safety profile in patients with LANPC. Trial Registration: ChiCTR240008603.
Keywords: PD‐1 inhibitor; chemotherapy; induction therapy; nasopharyngeal carcinoma; treatment response.
© 2025 The Author(s). Cancer Science published by John Wiley & Sons Australia, Ltd on behalf of Japanese Cancer Association.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures



Similar articles
-
Induction chemotherapy plus camrelizumab combined with concurrent chemoradiotherapy in locoregionally advanced nasopharyngeal carcinoma in non-endemic areas: a phase 2 clinical trial in North China.BMC Med. 2025 Feb 27;23(1):126. doi: 10.1186/s12916-025-03964-9. BMC Med. 2025. PMID: 40016735 Free PMC article. Clinical Trial.
-
Camrelizumab (SHR-1210) alone or in combination with gemcitabine plus cisplatin for nasopharyngeal carcinoma: results from two single-arm, phase 1 trials.Lancet Oncol. 2018 Oct;19(10):1338-1350. doi: 10.1016/S1470-2045(18)30495-9. Epub 2018 Sep 10. Lancet Oncol. 2018. PMID: 30213452 Clinical Trial.
-
Phase I/II dose-finding study of nanoparticle albumin-bound paclitaxel (nab®-Paclitaxel) plus Cisplatin as Treatment for Metastatic Nasopharyngeal Carcinoma.BMC Cancer. 2016 Jul 13;16:464. doi: 10.1186/s12885-016-2517-5. BMC Cancer. 2016. PMID: 27411683 Free PMC article. Clinical Trial.
-
Long-term results of locoregionally advanced nasopharyngeal carcinoma treated with cisplatin and 5-fluorouracil induction chemotherapy with or without docetaxel in young and middle aged adults.J Cancer Res Clin Oncol. 2025 Mar 4;151(3):99. doi: 10.1007/s00432-025-06145-6. J Cancer Res Clin Oncol. 2025. PMID: 40035865 Free PMC article.
-
Safety and Efficacy of Camrelizumab in Combination With Nab-Paclitaxel Plus S-1 for the Treatment of Gastric Cancer With Serosal Invasion.Front Immunol. 2022 Jan 18;12:783243. doi: 10.3389/fimmu.2021.783243. eCollection 2021. Front Immunol. 2022. PMID: 35116023 Free PMC article.
Cited by
-
Chemoradiotherapy Strategies for Immunotherapy-Sensitive Multi-Metastatic Nasopharyngeal Carcinoma: A Comparative Case Report and Literature Review.Curr Oncol. 2025 Aug 18;32(8):466. doi: 10.3390/curroncol32080466. Curr Oncol. 2025. PMID: 40862835 Free PMC article. Review.
References
-
- Li T., Li F., Guo X., et al., “Anti‐Epstein‐Barr Virus BNLF2b for Mass Screening for Nasopharyngeal Cancer,” New England Journal of Medicine 389, no. 9 (2023): 808–819. - PubMed
-
- Sun Y., Li W. F., Chen N. Y., et al., “Induction Chemotherapy Plus Concurrent Chemoradiotherapy Versus Concurrent Chemoradiotherapy Alone in Locoregionally Advanced Nasopharyngeal Carcinoma: A Phase 3, Multicentre, Randomised Controlled Trial,” Lancet Oncology 17, no. 11 (2016): 1509–1520. - PubMed
-
- Zhang Y., Chen L., Hu G. Q., et al., “Final Overall Survival Analysis of Gemcitabine and Cisplatin Induction Chemotherapy in Nasopharyngeal Carcinoma: A Multicenter, Randomized Phase III Trial,” Journal of Clinical Oncology 40, no. 22 (2022): 2420–2425. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

