The interstitial lung disease patient pathway: from referral to diagnosis
- PMID: 40040894
- PMCID: PMC11874298
- DOI: 10.1183/23120541.00899-2024
The interstitial lung disease patient pathway: from referral to diagnosis
Abstract
Background: Suspected interstitial lung disease (ILD) patients may be referred to an ILD-specialist centre or a non-ILD-specialist centre for diagnosis and treatment. Early referral and management of patients at ILD-specialist centres has been shown to improve survival and reduce hospitalisations. The COVID-19 pandemic has affected the ILD patient diagnostic pathway and prompted centres to adapt. This study investigates and contrasts ILD patient pathways in ILD-specialist and non-ILD-specialist centres, focusing on referrals, caseloads, diagnostic tools, multi-disciplinary team (MDT) meeting practices and resource accessibility.
Methods: Conducted as a cross-sectional study, a global self-selecting survey ran from September 2022 to January 2023. Participants included ILD specialists and healthcare professionals (HCPs) from ILD-specialist centres and non-ILD-specialist centres.
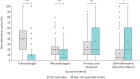
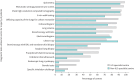
Results: Of 363 unique respondents from 64 countries, 259 were from ILD-specialist centres and 104 from non-ILD-specialist centres. ILD centres had better resource availability, exhibiting higher utilisation of diagnostic tests (median: 12 tests) than non-ILD centres (nine tests) and better access to specialist professions attending MDT meetings (median: six professions at meeting) in specialist centres than non-ILD centres (three professions at meeting). Transitioning to virtual MDT meetings allowed HCPs from other locations to join meetings in nearly 90% of all centres, increasing regular participation in 60% of specialist centres and 72% of non-ILD centres. For treatment of patients, specialist centres had better access to antifibrotic drugs (91%) compared to non-ILD centres (60%).
Conclusions: Diagnostic pathways for ILD patients diverged between specialist centres and non-ILD centres. Disparities in resource and specialist availability existed between centres.
Copyright ©The authors 2025.
Conflict of interest statement
Conflict of interest: G. Lough reports an unrestricted grant for the present study from Boehringer Ingelheim (BI). Conflict of interest: R. Abdulqawi has nothing to disclose. Conflict of interest: G. Amanda has nothing to disclose. Conflict of interest: K. Antoniou reports research grants from BI, Roche and Menarini; honoraria for lectures from BI, Roche, Elpen, Chiesi, GSK, Menarini and AstraZeneca; and travel grants from Chiesi, BI, Menarini and AstraZeneca, all in the past 36 months. Conflict of interest: A. Azuma reports consulting fees from Toray and Taiho; honoraria for manuscript writing from BI; and support for attending a steering committee meeting from BI, all in the past 36 months. Conflict of interest: M. Baldi has nothing to disclose. Conflict of interest: A. Bayoumy has nothing to disclose. Conflict of interest: J. Behr reports personal fees for lectures and consulting from AstraZeneca, Biogen, BI, Bristol Myers Squibb (BMS), Ferrer, Novartis and Sanofi Genzyme in the past 36 months. Conflict of interest: E. Bendstrup reports speaker's fees BI, Daichii-Sankyo, AstraZeneca and Chiesi; and advisory board fees from BI, Daichii-Sankyo and Veracyte, all in the past 36 months. Conflict of interest: D. Bouros has nothing to disclose. Conflict of interest: K. Brown reports serving as an external science advisor to AbbVie, AstraZeneca, CSL Behring, Dispersol, Eleven P15, Huitai Biomedicine and Redx Pharma; serving on data monitoring committees for Biogen, Humanetics and Scleroderma Research Foundation; serving on scientific advisory boards for Blade Therapeutics, BI, BMS, DevPro Pharma, Galapagos NV, Galecto, the Open Source Imaging Consortium (OSIC), Pliant, Sanofi and Trevi Therapeutics; serving as a consultant for Cumberland Pharma, Novartis and Shionogi; and leadership or fiduciary roles for the Fleischner Society and OSIC, all in the past 36 months. Conflict of interest: N. Chaudhuri has nothing to disclose. Conflict of interest: T.J. Corte reports institutional unrestricted educational grants from BI, Roche, Biogen, Galapagos, Pliant, 4D, Pharmaxis and Avalyn; consulting/advisory fees Roche, BMS, BI, Vicore, DevPro, Bridge Biotherapeutics, Ad Alta and Pliant; and speaker/travel fees from Roche, BI and BMS, all in the past 36 months. Conflict of interest: V. Cottin has nothing to disclose. Conflict of interest: B. Crestani has nothing to disclose. Conflict of interest: K.R. Flaherty reports grants or contracts from BI (paid to his institution); being a section editor of Up to Date; consulting around drug development and clinical trials in idiopathic pulmonary fibrosis and interstitial lung disease from Roche Genentech, Bellerophon, Respivant, Shionogi, DevPro, AstraZeneca, Pure, HealthHorizon, FibroGen, Sun Pharmaceuticals, Pliant, United Therapeutics, Arrowhead, Lupin, Polarean, PureTech, Trevi, CSL Behring, Daewoong, Dispersol, Immunet, NeRRe Therapeutics, Insilco, Vicore, GSK and Merck; and being a steering committee chair for the Pulmonary Fibrosis Foundation, in the past 36 months. Conflict of interest: I. Glaspole has nothing to disclose. Conflict of interest: L. Kawano-Dourado reports research grants from BI and BMS, and personal fees from BI and Roche, all in the past 36 months. Conflict of interest: M.P. Keane has nothing to disclose. Conflict of interest: M. Kolb reports BI provided funding for this project without input in data analysis and paper writing. He also reports research funding for preclinical work from BI, United Therapeutics and Structure Therapeutics; consulting fees from BI, Roche, Horizon, Cipla, Abbvie, Bellerophon, Algernon, CSL Behring, United Therapeutics, LabCorp, Structure Therapeutics, AstraZeneca, Pliant and Avalyn; payment of lectures, presentations, speakers’ bureaus, manuscript writing or educational events from Roche, Novarti and BI; payment for expert testimony from Roche; participation on a data safety monitoring or advisory board from United Therapeutics and Fortrea; and an allowance for serving as the Chief Editor of the European Respiratory Journal, all in the past 36 months. Conflict of interest: F.J. Martinez has nothing to disclose. Conflict of interest: M. Molina-Molina has nothing to disclose. Conflict of interest: I. Ojanguren has nothing to disclose. Conflict of interest: L. Pearmain has nothing to disclose. Conflict of interest: G. Raghu has nothing to disclose. Conflict of interest: P. Rottoli has nothing to disclose. Conflict of interest: S.C. Stanel has nothing to disclose. Conflict of interest: G. Tabaj has nothing to disclose. Conflict of interest: C. Vancheri has nothing to disclose. Conflict of interest: B. Varela reports personal fees from BI, Raffo, AstraZeneca, Bagó and Knight Therapeutics in the past 36 months. Conflict of interest: B. Wang has nothing to disclose. Conflict of interest: A. Wells has nothing to disclose. Conflict of interest: P. Rivera-Ortega has nothing to disclose.
Figures


References
-
- Vašáková M, Sterclova M, Kus J, et al. Does early diagnosis of idiopathic pulmonary fibrosis matter? Real-world's data from the EMPIRE registry. Eur Respir J 2016; 48: Suppl. 60, PA794. doi: 10.1183/13993003.congress-2016.PA794 - DOI
LinkOut - more resources
Full Text Sources
Miscellaneous
