Baseline Pathological Liver Function Tests in Patients With Psoriasis Support the Indication for Systemic Therapy Rather Than Being a Reason Against It: A Real-World Analysis
- PMID: 40046951
- PMCID: PMC11881625
- DOI: 10.2147/PTT.S502296
Baseline Pathological Liver Function Tests in Patients With Psoriasis Support the Indication for Systemic Therapy Rather Than Being a Reason Against It: A Real-World Analysis
Abstract
Purpose: Modern systemic therapies offer a high probability of significant improvement for psoriasis. However, elevated liver function tests (LFTs) may cause physicians to be reluctant to initiate systemic treatment. Especially considering the already increased risk of liver disease in patients with psoriasis, clinicians are often concerned about potential further liver damage caused by systemic therapies. The aim of this study was to provide real-world evidence to address this issue.
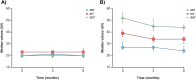
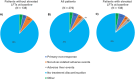
Patients and methods: This study retrospectively analyzed the treatment courses of N = 278 patients with psoriasis who received systemic anti-psoriatic therapy with secukinumab, ixekizumab, adalimumab, or apremilast in clinical routine. The cohort was divided into two subgroups based on normal or elevated LFTs prior to the start of therapy. AST, ALT, and GGT levels as well as Fibrosis-4 score (FIB-4) were measured at baseline, after 3 months, and after 6 months of therapy.
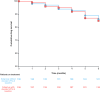
Results: The subgroup of patients with elevated LFTs had a higher mean PASI (Psoriasis Area and Severity Index), were more likely to be male, and had a higher prevalence of metabolic syndrome comorbidities compared to the subgroup with normal LFTs. During the follow-up period, there were no significant changes in LFTs and FIB-4 for the subgroup with normal LFTs at baseline. In the group of patients with initially elevated LFTs, all LFTs decreased significantly over time, whereas FIB-4 scores demonstrated no significant change. Drug survival, discontinuation rates, and PASI-75 response did not significantly differ between subgroups.
Conclusion: This study provides real world evidence that systemic therapy with secukinumab, ixekizumab, adalimumab, or apremilast does not present a general risk, but rather an opportunity for patients with psoriasis with elevated LFTs at baseline.
Keywords: biologics; hepatic dysfunction; psoriasis; real world data; safety.
© 2025 Krefting et al.
Conflict of interest statement
Frederik Krefting received travel support for participation in congresses and / or (speaker) honoraria from Novartis, Lilly, Bristol-Myers Squibb, Janssen, Pierre Fabre, Almirall, and Boehringer Ingelheim outside of the present publication. Sven Benson reports grants from German Research Foundation (DFG), during the conduct of the study; received speaker honoraria from Astra Zeneca, Bayer, Celgene, Grünenthal, Janssen-Cilag, Lilly, Novo Nordisk, Sanofi-Aventis, Symbiopharm, and Urgo outside of the present publication. Heiner Wedemeyer is on the speakers’ bureau for, received grants from, and has other interests in Gilead Sciences, Inc. He consults for, received grants from, and has other interests in Falk. He consults for and has other interests in Merck Sharp & Dohme. He consults for and advises Roche, Abbott Laboratories & Abbott Molecular Inc., Albireo Pharma Inc., AstraZeneca GmbH, Atea Pharmaceuticals, Inc., F. Hoffmann-La Roche Ltd., Orphalan GmbH, Pfizer Pharma GmbH, Roche Diagnostics International Ltd., Roche Pharma AG, Sobi - Swedish Orphalan Biovitrum GmbH, Takeda Pharma Vertrieb GmbH & Co. KG, and Vir Biotechnology, Inc. He consults for and is on the speakers’ bureau for Pfizer Inc, BioMarin Pharmaceuticals Inc. and CSL Behring. He is on the speakers’ bureau for the Falk Foundation and Olink. Wiebke Sondermann received travel support for participation in congresses and / or (speaker) honoraria as well as research grants from medi GmbH Bayreuth, AbbVie, Almirall, Amgen, Boehringer Ingelheim, Bristol-Myers Squibb, Celgene, GSK, Janssen, LEO Pharma, Lilly, MSD, Novartis, Pfizer, Roche, Sanofi Genzyme, and UCB outside of the present publication. Jan-Malte Placke, Stefanie Hölsken, and Cosima Scheib declare no conflicts of interest in this work.
Figures

References
LinkOut - more resources
Full Text Sources
Miscellaneous

