Podoplanin-positive cell-derived small extracellular vesicles contribute to cardiac amyloidosis after myocardial infarction
- PMID: 40056419
- PMCID: PMC12019684
- DOI: 10.1016/j.celrep.2025.115408
Podoplanin-positive cell-derived small extracellular vesicles contribute to cardiac amyloidosis after myocardial infarction
Abstract
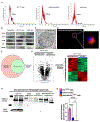
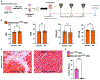
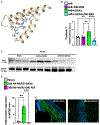
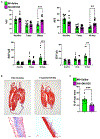
Cardiac amyloidosis is a secondary phenomenon of an already pre-existing chronic condition. Whether cardiac amyloidosis represents one of the complications post myocardial infarction (MI) has yet to be fully understood. Here, we show that amyloidosis occurs after MI and that amyloid fibers are composed of macrophage-derived serum amyloid A 3 (SAA3) monomers. SAA3 overproduction in macrophages is triggered by exosomal communication from cardiac stromal cells (CSCs), which, in response to MI, activate the expression of a platelet aggregation-inducing type I transmembrane glycoprotein, Podoplanin (PDPN). CSCPDPN+-derived small extracellular vesicles (sEVs) are enriched in SAA3, and exosomal SAA3 engages with macrophage by Toll-like receptor 2, triggering overproduction with consequent impaired clearance and aggregation of SAA3 monomers into rigid fibers. SAA3 amyloid deposits reduce cardiac contractility and increase scar stiffness. Inhibition of SAA3 aggregation by retro-inverso D-peptide, specifically designed to bind SAA3 monomers, prevents the deposition of SAA3 amyloid fibrils and improves heart function post MI.
Keywords: CP: Cell biology; CP: Metabolism; Podoplanin; Toll-like receptor 2; cardiac amyloidosis; myocardial infarction; retro-inverso D-peptide; serum amyloid A 3.
Copyright © 2025 The Author(s). Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
Declaration of interests The authors declare no competing interests.
Figures



Update of
-
Podoplanin Positive Cell-derived Extracellular Vesicles Contribute to Cardiac Amyloidosis After Myocardial Infarction.bioRxiv [Preprint]. 2024 Jul 2:2024.06.28.601297. doi: 10.1101/2024.06.28.601297. bioRxiv. 2024. Update in: Cell Rep. 2025 Mar 25;44(3):115408. doi: 10.1016/j.celrep.2025.115408. PMID: 39005419 Free PMC article. Updated. Preprint.
References
-
- Pagourelias ED, Mirea O, Duchenne J, Van Cleemput J, Delforge M, Bogaert J, Kuznetsova T, and Voigt JU (2017). Echo Parameters for Differential Diagnosis in Cardiac Amyloidosis: A Head-to-Head Comparison of Deformation and Nondeformation Parameters. Circ. Cardiovasc. Imaging 10, e005588. 10.1161/CIRCIMAGING.116.005588. - DOI - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Miscellaneous

