Safety and Efficacy of DTX401, an AAV8-Mediated Liver-Directed Gene Therapy, in Adults With Glycogen Storage Disease Type I a (GSDIa)
- PMID: 40064185
- PMCID: PMC11893205
- DOI: 10.1002/jimd.70014
Safety and Efficacy of DTX401, an AAV8-Mediated Liver-Directed Gene Therapy, in Adults With Glycogen Storage Disease Type I a (GSDIa)
Abstract
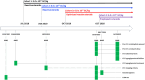
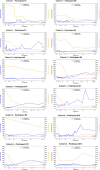
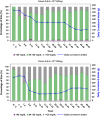
Glycogen storage disease type Ia (GSDIa) is a rare, life-threatening, inherited carbohydrate metabolism disorder caused by glucose-6-phosphatase (G6Pase) deficiency, which is essential for glycogenolysis and gluconeogenesis. GSDIa management includes a strict medically prescribed diet that typically includes daily uncooked cornstarch doses, including overnight, to maintain euglycemia. DTX401 is an investigational adeno-associated virus serotype 8 vector expressing the human G6PC1 gene that encodes G6Pase. This open-label, phase 1/2, dose-escalation, 52-week gene therapy trial evaluated the safety and efficacy of a single DTX401 infusion in 12 adults with GSDIa (ClinicalTrials.gov Identifier: NCT03517085). Three participants in Cohort 1 received DTX401 2.0 × 1012 genome copies (GC)/kg, and three participants each in Cohorts 2, 3, and 4 received 6.0 × 1012 GC/kg. Corticosteroids were administered to mitigate vector‑induced inflammatory response. All participants experienced a treatment-emergent adverse event (TEAE) and a related TEAE. No participant experienced a dose-limiting toxicity, TEAE leading to study discontinuation, TEAE leading to death, or serious treatment-related TEAE. Mean (SD) time to hypoglycemia in minutes/gram of carbohydrate during a controlled fasting challenge was 5.0 (1.6) at baseline and 6.9 (2.7) at Week 52, a mean (SD) increase of 46% (72%). Mean total daily cornstarch intake was 284 g at baseline and 85 g at Week 52 in the 10 participants with available values at both time points, a mean (SD) total daily cornstarch intake reduction of 68% (20%); p < 0.001. DTX401 showed a favorable safety and efficacy profile at Week 52. Participants in all cohorts showed significant cornstarch need reductions from baseline to Week 52.
Keywords: DTX401; GSD; GSDIa; gene therapy; glycogen storage disease.
© 2025 The Author(s). Journal of Inherited Metabolic Disease published by John Wiley & Sons Ltd on behalf of SSIEM.
Conflict of interest statement
For all authors, Ultragenyx provided the investigational agent, grant support to their institutions for the conduct of this clinical trial, and medical writing support in the preparation of this manuscript. David A. Weinstein served on the scientific advisory board for Dimension Therapeutics prior to the acquisition of Dimension by Ultragenyx and is an unpaid member of the board of directors of the Association for Glycogen Storage Disease. Terry G. Derks reported financial research support for investigator‐initiated research and sponsor‐initiated research from Ultragenyx, consulting fees from Danone, Ultragenyx, ModernaTX Inc., and Beam Therapeutics, honoraria for lectures or presentations on behalf of MEDTalks, Prelum, Danone, and participation on a data safety monitoring board/advisory board for ModernaTX Inc. David F. Rodriguez‐Buritica received consulting fees from Ultragenyx, Moderna, and Beam Therapeutics. John J. Mitchell received consulting fees from Biomarin, Takeda, and Sanofi‐Genzyme, payment or honoraria from Biomarin and Takeda, and participated on a data monitoring committee for Moderna. Rebecca Riba‐Wolman received consulting fees from Beam Therapeutics and payment or honoraria from Ultragenyx. Malaya Mount received consulting fees from ModernaTX Inc, Beam Therapeutics, and Danone, as well as travel support for attending meetings from ModernaTX Inc and Danone. Heather Saavedra received consulting fees from Danone (paid to institution), ModernaTX Inc., Beam Therapeutics, Sanofi, Vitaflo International, and payment or honoraria and support for attending meetings from Ultragenyx. Deepali Mitragotri is an employee and shareholder of Ultragenyx. Eric Crombez is an employee and shareholder of Ultragenyx. Ayesha Ahmad, María‐Luz Couce, Julieta Bonvin Sallago, Katalin M. Ross, Melanie M. van der Klauw, Foekje de Boer, Caroline van der Schaaf, Miguel Martínez‐Olmos, Elvis Atanga, and Asad Hosseini authors declare no conflicts of interest.
Figures


References
-
- Bali D. S., El‐Gharbawy A., Austin S., Pendyal S., and Kishnani P. S.. 2006. “Glycogen Storage Disease Type I,” In GeneReviews® [Internet], edited by Adam M. P., Everman D. B., Mirzaa G. M., et al. University of Washington. - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

