Ginseng exosomes modulate M1/M2 polarisation by activating autophagy and target IKK/IкB/NF-кB to alleviate inflammatory bowel disease
- PMID: 40065319
- PMCID: PMC11895377
- DOI: 10.1186/s12951-025-03292-3
Ginseng exosomes modulate M1/M2 polarisation by activating autophagy and target IKK/IкB/NF-кB to alleviate inflammatory bowel disease
Abstract
Background: Exosomes are involved in intercellular communication and regulation of the inflammatory microenvironment. In a previous study, we demonstrated that fresh ginseng exosomes (GEs) alleviated inflammatory bowel disease. However, the precise mechanism by which GEs activate the immune system and subsequently inhibit the formation of intestinal inflammatory microenvironment remains unknown.
Methods: Herein, we investigated the effects of GEs on autophagy, macrophage polarisation, intestinal inflammation, and the epithelial barrier by means of transcriptome sequencing, network pharmacology, transmission electron microscopy, immunoblotting, flow cytometry and small molecule inhibitors.
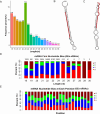
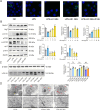
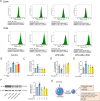
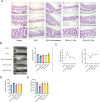
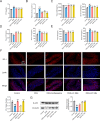
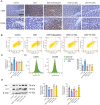
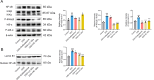
Results: GEs significantly activated autophagy and M2-like macrophage polarisation, which could be blocked by the autophagy inhibitor 3-methyladenine. In the co-culture system of macrophages and intestinal epithelial cells, macrophages treated with GEs secreted more interleukin-10 (IL-10) and significantly reduced Nitric oxide (NO) levels in intestinal epithelial cells in vitro. Furthermore, GEs acted directly on intestinal epithelial cells through the IKK/IкB/NF-кB signalling pathway to reduce inflammation and restore the intestinal barrier. Orally administered GEs could restore disrupted colonic barriers, alleviate inflammatory bowel responses, and regulate the polarisation of intestinal macrophages in vivo.
Conclusion: In summary, GEs may be a potential treatment for inflammatory bowel disease, and targeting autophagy and macrophage polarisation may help alleviate intestinal inflammation.
Keywords: Autophagy; Ginseng exosomes; Intestinal barrier; M1/M2 polarisation.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Ethics approval and consent to participate: Animal studies were approved by the Laboratory Animal Ethics Committee of Changchun University of Traditional Chinese Medicine (No. 2024382). Consent for publication: All authors read and agreed to submit the manuscript. Competing interests: The authors declare no competing interests.
Figures



Similar articles
-
Exosomal lncRNA GAS5 promotes M1 macrophage polarization in allergic rhinitis via restraining mTORC1/ULK1/ATG13-mediated autophagy and subsequently activating NF-кB signaling.Int Immunopharmacol. 2023 Aug;121:110450. doi: 10.1016/j.intimp.2023.110450. Epub 2023 Jun 19. Int Immunopharmacol. 2023. PMID: 37343372
-
Ginseng root extract attenuates inflammation by inhibiting the MAPK/NF-κB signaling pathway and activating autophagy and p62-Nrf2-Keap1 signaling in vitro and in vivo.J Ethnopharmacol. 2022 Jan 30;283:114739. doi: 10.1016/j.jep.2021.114739. Epub 2021 Oct 11. J Ethnopharmacol. 2022. PMID: 34648903
-
MCP-1-induced protein attenuates endotoxin-induced myocardial dysfunction by suppressing cardiac NF-кB activation via inhibition of IкB kinase activation.J Mol Cell Cardiol. 2011 Aug;51(2):177-86. doi: 10.1016/j.yjmcc.2011.04.018. Epub 2011 May 17. J Mol Cell Cardiol. 2011. PMID: 21616078
-
[Participation of heat shock proteins in modulation of NF-кB transcription factor activation during bacterial infections].Postepy Hig Med Dosw (Online). 2015 Jan 2;69:969-77. doi: 10.5604/17322693.1165199. Postepy Hig Med Dosw (Online). 2015. PMID: 26400883 Review. Polish.
-
Development, validation and implementation of an in vitro model for the study of metabolic and immune function in normal and inflamed human colonic epithelium.Dan Med J. 2015 Jan;62(1):B4973. Dan Med J. 2015. PMID: 25557335 Review.
Cited by
-
Quercetin improves myocardial ischemia-reperfusion injury by regulating macrophage M2 polarization through Bcl-2/Beclin-1 complex.Eur J Med Res. 2025 Aug 21;30(1):780. doi: 10.1186/s40001-025-03077-2. Eur J Med Res. 2025. PMID: 40836312 Free PMC article.
-
Mesenchymal stem cell-derived apoptotic vesicles regulate irritable bowel syndrome in mice via the 5-HT brain-gut axis.Stem Cell Res Ther. 2025 Jul 1;16(1):326. doi: 10.1186/s13287-025-04448-6. Stem Cell Res Ther. 2025. PMID: 40598603 Free PMC article.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

