Bowel Disorder Incidence and Rectal Spacer Use in Patients With Prostate Cancer Undergoing Radiotherapy
- PMID: 40067300
- PMCID: PMC11897833
- DOI: 10.1001/jamanetworkopen.2025.0491
Bowel Disorder Incidence and Rectal Spacer Use in Patients With Prostate Cancer Undergoing Radiotherapy
Abstract
Importance: The polyethylene glycol-based hydrogel spacer (PHS) system temporarily separates the rectum from the prostate in patients undergoing radiotherapy (RT) for prostate cancer (PCa).
Objective: To compare incidence of bowel disorders and related procedures in patients receiving RT with and without PHS.
Design, setting, and participants: This retrospective cohort study used 4 datasets: Medicare 5% Standard Analytic Files, Medicare 100% Standard Analytic Files, Merative MarketScan Commercial Database, and Premier Healthcare Database. Participants included adult patients with PCa undergoing RT from 2015 to 2021.
Exposure: Placement of PHS.
Main outcomes: All-cause bowel disorders and related procedures, identified from diagnosis and procedure codes. Results were compared with age-matched male general population without PCa or RT.
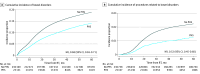
Results: Of 261 906 patients with PCa included in the study, 25 167 (9.6%) received PHS (mean [SD] age, 70.7 [6.5] years) and 236 739 did not (mean [SD] age, 71.1 [7.5] years). One year prior to RT, patients who received PHS had a lower mean (SD) Charlson Comorbidity Index score than those who did not (2.48 [1.08] vs 3.14 [1.95]; P < .001). Stereotactic RT was more common in patients who received PHS (2743 [10.9%] vs 8810 [3.7%]; P < .001), while intensity-modulated RT was less common (12 755 [50.7%] vs 142 402 [60.2%]; P < .001). After 4 years post RT, patients who received PHS had a 25% lower hazard of bowel disorders (hazard ratio [HR], 0.75 [95% CI, 0.72-0.78]; P < .001) and a 46% lower hazard of related procedures (HR, 0.54 [95% CI, 0.47-0.62]; P < .001) than patients who did not receive PHS. Patients without PHS had higher hazard compared with an age-matched general population (disorders: 17.1% [95% CI, 17.3%-17.6%] vs 10.3% [95% CI, 10.1%-10.5%]; HR, 1.35 [95% CI, 1.32-1.37]; P < .001; procedures: 2.0% [95% CI, 1.9%-2.1%] vs 0.7% [95% CI, 0.7%-0.8%]; HR, 1.92 [95% CI, 1.79-2.06]; P < .001), while patients who received PHS did not (disorders: 12.4% vs 10.3%; HR, 1.00 [95% CI, 0.98-1.05]; P = .82; procedures: 1.1% [95% CI, 1.0%-1.3%] vs 0.7% [95% CI, 0.7%-0.8%]; HR, 1.11 [95% CI, 0.96-1.29]; P = .15). Common procedures included colonoscopy, sigmoidoscopy, and rectal resection.
Conclusions and relevance: In this cohort study of patients with PCa receiving RT, those receiving a PHS had a significantly lower incidence of all-cause bowel disorders and related procedures compared with patients who did not receive a PHS over the 4-year follow-up. The incidence among patients with PHS was similar to the general population. These findings are consistent with prior phase 3 trial results, where patients receiving PHS experienced no decline in bowel quality of life.
Conflict of interest statement
Figures
References
-
- American Cancer Society . Key statistics for prostate cancer. 2024; Accessed May 17, 2024. https://www.cancer.org/cancer/types/prostate-cancer/about/key-statistics...
-
- Bekelman JE, Rumble RB, Chen RC, et al. . Clinically Localized Prostate Cancer: ASCO Clinical Practice Guideline Endorsement of an American Urological Association/American Society for Radiation Oncology/Society of Urologic Oncology Guideline. J Clin Oncol. 2018;36(32):3251-3258. doi:10.1200/JCO.18.00606 - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical