Enhancing MyD88 oligomerization is one important mechanism by which IBDV VP2 induces inflammatory response
- PMID: 40067802
- PMCID: PMC11957393
- DOI: 10.1371/journal.ppat.1012985
Enhancing MyD88 oligomerization is one important mechanism by which IBDV VP2 induces inflammatory response
Abstract
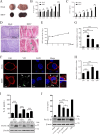
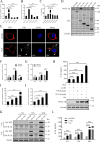
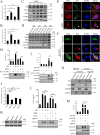
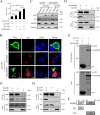
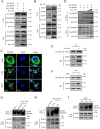
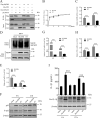
The inflammatory response is an essential component of innate immunity to defense against pathogens. Infectious bursal disease (IBD) is the most important immunosuppressive disease in chickens and is caused by the infectious bursal disease virus (IBDV). Acute inflammation is a typical pathogenic process for IBD, however, the underlying mechanism is not clear. Here, we report that IBDV induces obvious inflammatory response in vivo and in vitro. Furthermore, viral VP2 is identified as an important inflammatory stimulus. It is observed that IBDV VP2 can activate NF-κB signaling pathway and then increase IL-1β production. In detail, IBDV VP2 interacts with myeloid differentiation primary response gene 88 (MyD88), potentiates the oligomerization of MyD88 and assembly of MyD88 complex, which is one important element leading to NF-κB signaling pathway activation and IL-1β production increase. More meaningfully, residues 253/284 of viral VP2 are significantly involved in IBDV-induced inflammatory response through modulating the interaction strength between VP2 and MyD88 and the following MyD88-NF-κB-IL-1β signaling pathway. This study reveals one molecular mechanism that trigger inflammation during IBDV infection, which is of great significance for a deeper understanding of the pathogenic mechanisms of IBDV.
Copyright: © 2025 Huang et al. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures

References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

