Mesenchymal stem cell-derived extracellular vesicles alleviate autism by regulating microglial glucose metabolism reprogramming and neuroinflammation through PD-1/PD-L1 interaction
- PMID: 40069859
- PMCID: PMC11895333
- DOI: 10.1186/s12951-025-03250-z
Mesenchymal stem cell-derived extracellular vesicles alleviate autism by regulating microglial glucose metabolism reprogramming and neuroinflammation through PD-1/PD-L1 interaction
Abstract
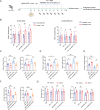
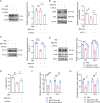
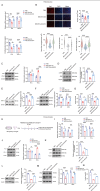
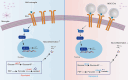
Neuroinflammation triggered by microglia activation is hallmark of autism spectrum disorder (ASD), and this process includes crucial metabolic reprogramming from oxidative phosphorylation to glycolysis, which may cause neuron loss and functional impairment. The inhibitory immune checkpoint programmed cell death protein 1 (PD-1) on immune cells is an important target for tumor immunotherapy. However, the immunomodulatory effects of PD-1 in ASD remains to be elusive. Mesenchymal stem cell-derived extracellular vesicles (MSC-EVs) exhibit immunomodulatory capabilities in a range of neurological diseases. Our findings indicated the expression of PD-L1 on MSC-EVs, potentially facilitating signaling to PD-1-expressing microglia. Here, we showed how MSC-EVs activated of PD-L1/PD-1 axis and ameliorated glycolysis, neuroinflammation and autism-like behaviors. After first detecting elevated glycolysis and neuroinflammation in prefrontal cortex (PFC) tissue from the maternal immune activation (MIA) mice, we also demonstrated that PD-1 expression level was upregulated in microglia. Following given MSC-EVs carried PD-L1 into adult MIA offspring mice via intranasal administration, which bound with PD-1 on microglia and then the autism-like behaviors were alleviated as well. Further experiments verified that MSC-EVs could decreased the level of glycolysis and neuroinflammation by PD-1/ERK/HIF-1α pathway in the primary microglia in PFC of MIA offspring mice. Pharmacological blockade and genetic inhibition of PD-1 could weaken the effect of MSC-EVs and aggravate microglial dysfunction, glycolysis and autism-like behaviors in MIA offspring mice. Futhermore, PD-L1 deficient weakened the effect of MSC-EVs on neuroinflammation, glycolysis and autism-like behaviors in MIA offspring mice. Our research indicated the significant immunomodulatory capabilities of MSC-EVs, which play an important role in reprogramming microglial glucose metabolism and suppressing neuroinflammation in ASD. By activating the PD-L1/PD-1 axis and inhibiting the downstream ERK/HIF-1α pathway, MSC-EVs were found to alleviate autism-like behaviors, which revealing a novel pathological mechanism and offering promising therapeutic insights into ASD.
Keywords: Autism spectrum disorder; Glycolysis; MSC-EVs; Neuroinflammation; PD-L1/PD-1.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Ethics approval and consent to participate: The mouse experiments were approved by the Ethics Committee of Harbin Medical University (HMUIRB2023015). Consent for publication: All authors agree with the publication of this paper. Competing interests: The authors declare no competing interests.
Figures










References
-
- Maenner MJ, Warren Z, Williams AR, Amoakohene E, Bakian AV, Bilder DA, Durkin MS, Fitzgerald RT, Furnier SM, Hughes MM, et al. Prevalence and characteristics of autism spectrum disorder among children aged 8 Years - autism and developmental disabilities monitoring network, 11 sites, united States, 2020. MMWR Surveill Summ. 2023;72:1–14. - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

