Chronic Pain Induced by Social Defeat Stress in Juvenile Mice Depends on TLR4
- PMID: 40072079
- PMCID: PMC11898947
- DOI: 10.3390/cells14050350
Chronic Pain Induced by Social Defeat Stress in Juvenile Mice Depends on TLR4
Abstract
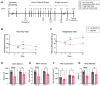
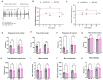
A significant portion of adolescents suffer from mental illnesses and persistent pain due to repeated stress. The components of the nervous system that link stress and pain in early life remain unclear. Prior studies in adult mice implicated the innate immune system, specifically Toll-like receptors (TLRs), as critical for inducing long-term anxiety and pain-like behaviors in social defeat stress (SDS) models. In this work, we investigated the pain and anxiety behavioral phenotypes of wild-type and TLR4-deficient juvenile mice subjected to repeated SDS and evaluated the engagement of TLR4 by measuring dimerization in the spinal cord, dorsal root ganglia, and prefrontal cortex. Male juvenile (4-week-old) mice (C57BL/6J or Tlr4-/-) underwent six social defeat sessions with adult aggressor (CD1) mice. In WT mice, SDS promotes chronic mechanical allodynia and thermal hyperalgesia assessed via von Frey testing and the Hargreaves test, respectively. In parallel, the stressed WT mice exhibited transient anxiety-like behavior and long-lasting locomotor activity reduction in the open-field test. Tlr4-/--stressed animals were resistant to the induction of pain-like behavior but had a remnant of anxious behavior, spending less time in the center of the arena. In WT SDS, there were concordant robust increases in TLR4 dimerization in dorsal root ganglia macrophages and spinal cord microglia, indicating TLR4 activation. These results suggest that the chronic pain phenotype and locomotor impairment induced by SDS in juvenile mice depends on TLR4 engagement evidenced by dimerization in immune cells of the dorsal root ganglia and spinal cord.
Keywords: TLR4; dorsal root ganglia; pain; social stress; spinal cord.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures


References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

