Impact of treatment for adolescent and young adults with essential thrombocythemia and polycythemia vera
- PMID: 40074852
- PMCID: PMC12055580
- DOI: 10.1038/s41375-025-02545-2
Impact of treatment for adolescent and young adults with essential thrombocythemia and polycythemia vera
Abstract
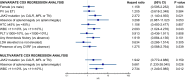
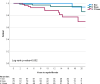
Essential thrombocythemia (ET) and polycythemia vera (PV) are rare in adolescent and young adult (AYA). These conditions, similar to those in older patients, are linked with thrombotic complications and the potential progression to secondary myelofibrosis (sMF). This retrospective study of ET and PV patients diagnosed before age 25 evaluated complication rates and impact of cytoreductive drugs on outcomes. Among 348 patients (278 ET, 70 PV) with a median age of 20 years, the of thrombotic events was 1.9 per 100 patient-years. Risk factors for thrombosis included elevated white blood cell count (>11 × 109/L) (HR: 2.7, p = 0.012) and absence of splenomegaly at diagnosis (HR: 5.7, p = 0.026), while cytoreductive drugs did not reduce this risk. The incidence of sMF was 0.7 per 100 patient-years. CALR mutation (HR: 6.0, p < 0.001) and a history of thrombosis (HR: 3.8, p = 0.015) were associated with sMF risk. Interferon as a first-line treatment significantly improved myelofibrosis-free survival compared to other treatments or the absence of cytoreduction (p = 0.046). Although cytoreduction did not affect thrombotic event, early interferon use reduced sMF risk. These findings support interferon use to mitigate sMF risk in AYA ET and PV patients.
© 2025. The Author(s).
Conflict of interest statement
Competing interests: YB reports advisory board membership (AOP), speaker fee (AOP), congress support (Abbvie), J-CI reports advisory board membership (Abbvie, Novartis), grant (Novartis), congresses support (Novartis, Pfizer, GSK), MS reports lectures (AOP Orphan, BMS, GSK, Novartis), conferences (Novartis), membership advisory board (BMS, GSK), TD reports consultancy fee (GSK, Morphosys, BMS, Novartis, Incyte) and honorarium (Janssen, Sobi, Alexion), MFM reports advisory board membership (Novartis, BMS, Incyte, GSK), speaker bureau participation (Novartis, AOP, GSK), and clinical trial support (BMS, AOP, GSK), LL reports research funding (Amgen, BMS, Incyte, Novartis), honoraria (BMS, GSK, Novartis, Pfizer), hospitality (Novartis, Pfizer), FG reports sponsored UPR (Abbvie), CJ reports conference (Abbvie), MW reports speaker fees (AOP), educational talks (Novartis, BMS, Sanofi), data safety monitoring board (Keros therapeutics), MG report consultancy (AOP Orphan, Novartis, BMS, AbbVie, Pfizer, Roche, Janssen, Gilead, AstraZeneca, Sierra, Lilly, GSK) and honorarium (AOP Orphan, Novartis, BMS, AbbVie, Pfizer, Roche, Janssen, Gilead, AstraZeneca, Sierra, Lilly, GSK), J-JK reports consultancy fee (Novartis, GSK, Abbvie), advisory boards membership (BMS, Incyte, AOP, PharmaEssentia), CNH reports research funding (Celgene (BMS), Constellation, GSK, Novartis), and Advisory board membership (AbbVie, AOP, BMS, CTI, IMAGO, Incyte, Novartis, Galacteo, Geron, GSK, Incyte, Janssen, Keros, MSD, SOBI, Morphosys). The remaining authors declare no competing financial interests.
Figures



References
-
- Arber DA, Orazi A, Hasserjian R, Thiele J, Borowitz MJ, Le Beau MM, et al. The 2016 revision to the World Health Organization classification of myeloid neoplasms and acute leukemia. Blood. 2016;127:2391–405. - PubMed
-
- Tefferi A, Barbui T. Polycythemia vera: 2024 update on diagnosis, risk-stratification, and management. Am J Hematol. 2023;98:1465–87. - PubMed
-
- Szuber N, Vallapureddy RR, Penna D, Lasho TL, Finke C, Hanson CA, et al. Myeloproliferative neoplasms in the young: Mayo Clinic experience with 361 patients age 40 years or younger. Am J Hematol. 2018;93:1474–84. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

