Sympathetic and parasympathetic subtypes of body-first Lewy body disease observed in postmortem tissue from prediagnostic individuals
- PMID: 40082617
- PMCID: PMC12081295
- DOI: 10.1038/s41593-025-01910-9
Sympathetic and parasympathetic subtypes of body-first Lewy body disease observed in postmortem tissue from prediagnostic individuals
Abstract
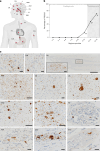
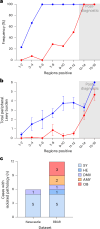
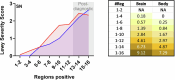
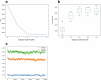
Recent studies suggest the existence of brain-first and body-first subtypes within the Lewy body disorder (LBD) spectrum, including Parkinson's disease. These studies primarily focused on α-synuclein propagation through the parasympathetic vagal and olfactory bulb routes, leaving the possibility of a sympathetic nervous system spreading route unexplored. In the present study, we analyzed two postmortem datasets, which included 173 and 129 cases positive for Lewy pathology. We observed a clear distinction between brain-first and body-first subtypes in early prediagnostic cases with mild Lewy pathology. Brain-first cases displayed minimal peripheral organ pathology in prediagnostic phases, contrasting with marked autonomic involvement in prediagnostic body-first cases. Utilizing the SuStaIn machine learning algorithm, we identified two distinct body-first subtypes, one with vagal predominance and another with sympathetic predominance, in equal proportions. Our study supports the existence of three prediagnostic LBD subtypes and highlights the sympathetic nervous system alongside the parasympathetic system in LBD onset and progression.
© 2025. The Author(s).
Conflict of interest statement
Competing interests: J.B.T. has served as a speaker for EISAI and a consultant for GE Healthcare. P.B. holds grants from the Lundbeck foundation, Danish Research Council, Parkinsonforeningen and Jaschafonden, has served as a speaker for EISAI and AbbVie and as a consultant for Novo Nordisk, Curium, AbbVie and Roche. Y.S. and S.M. hold grants from JSPS KAKENHI (grant no. JP22H04923; CoBiA), Integrated Research Initiative for Living Well with Dementia (IRIDE) of the Tokyo Metropolitan Institute for Geriatrics and Gerontology IRIDE, AMED (grant no. JP21wm0425019). Y.S. holds a grant from MHLW Research on the rare and intractable diseases program (grant no. JPMH23FC1008). J.W.V. was supported by the SciLifeLab & Wallenberg Data Driven Life Science Program (grant no. KAW 2020.0239). J.H. has received a speaker’s fee from AbbVie and grant support from the Lundbeck foundation. N.V.D.B. holds research grants from Lundbeck Foundation and the Danish Medical Research Council. The authors declare that these research grants, appointments and activities do not constitute competing interests for the current work. The other authors declare no competing interests.
Figures






References
MeSH terms
Grants and funding
- R359-2020-2533/Lundbeckfonden (Lundbeck Foundation)
- 3165-00212A/Sundhed og Sygdom, Det Frie Forskningsråd (Medical Sciences, Danish Council for Independent Research)
- None/Jascha Fonden (Jascha Foundation)
- A901/Parkinsonforeningen (Danish Parkinson Association)
- KAW 2020.0239/Science for Life Laboratory (SciLifeLab)
LinkOut - more resources
Full Text Sources
Medical

