Target inhibition of SPAK in choroid plexus attenuates T cell infiltration and demyelination in experimental autoimmune encephalomyelitis
- PMID: 40082912
- PMCID: PMC11907836
- DOI: 10.1186/s12974-025-03407-5
Target inhibition of SPAK in choroid plexus attenuates T cell infiltration and demyelination in experimental autoimmune encephalomyelitis
Abstract
Background: Disease-modifying therapies (DMTs) that prevent immune cell infiltration into the brain have demonstrated efficacy in multiple sclerosis (MS) treatment. However, their unpredictable adverse effects necessitate the development of safer therapeutic alternatives. The choroid plexus (ChP) functions as a crucial barrier against immune cell invasion, and previous studies have shown that preventing immune cell infiltration across the ChP reduces brain lesion in MS animal models. Understanding ChP barrier regulation is therefore essential for identifying novel therapeutic targets for MS. Here, we explored the role of Ste20-related proline/alanine-rich kinase (SPAK) in experimental autoimmune encephalomyelitis (EAE).
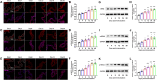
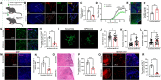
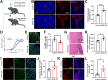
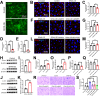
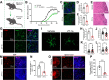
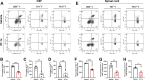
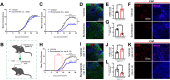
Methods: We examined the expression patterns of SPAK signaling in ChP using immunofluorescence in the EAE model. To investigate the roles of SPAK, matrix metalloproteinase (MMP) 2 and MMP9 in EAE pathology, we performed ChP-specific gene manipulation via intracerebroventricular (ICV) injection of recombinant adeno-associated virus 2/5 (rAAV2/5). T cell infiltration into the central nervous system (CNS) was analyzed using CD4 immunostaining and flow cytometry. We employed cell immunofluorescence, transwell assays, and rescue experiments in vitro to study SPAK's effects on ChP epithelial barrier integrity. We also evaluated the protective effects of SPAK-Na-K-2Cl cotransporter-1 (NKCC1) inhibitors (ZT-1a and bumetanide) on immune invasion and demyelination during EAE using pharmacological approaches.
Results: Following EAE induction, we observed progressive increases in both total and phosphorylated SPAK levels in ChP epithelium. Notably, ChP-specific SPAK knockdown significantly reduced T cell invasion and ameliorated EAE pathology, while SPAK overexpression exacerbated these effects. Bulk RNA sequencing and subsequent qPCR validation revealed that SPAK knockdown decreased the expression of MMP2 and MMP9, MMPs that compromise barrier integrity by degrading tight junction proteins. In vitro studies demonstrated that SPAK overexpression impaired ChP barrier function through the activator protein-1 (AP-1)-MMP2/9-zonula occludens-1 (ZO-1) axis. Furthermore, ChP-specific knockdown of either MMP2 or MMP9 protected against EAE pathology. Additionally, we identified SPAK-NKCC1 antagonists (bumetanide and ZT-1a) as promising therapeutic candidates for MS/EAE treatment.
Conclusions: Our findings demonstrate that targeting ChP-SPAK signaling represents a novel therapeutic strategy for MS treatment.
Keywords: Choroid plexus; Experimental autoimmune encephalomyelitis; Multiple sclerosis; SPAK; Tight junctions.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Ethics approval and consent to participate: The study (ethics protocol number: HT-2024-LWFB-0022) was approved by the Animal Care and Use Committee of Zhejiang Huitong Test & Evaluation Technology Group. Consent for publication: Not applicable. Competing interests: The authors declare no competing interests.
Figures



Similar articles
-
Role of SPAK-NKCC1 signaling cascade in the choroid plexus blood-CSF barrier damage after stroke.J Neuroinflammation. 2022 Apr 12;19(1):91. doi: 10.1186/s12974-022-02456-4. J Neuroinflammation. 2022. PMID: 35413993 Free PMC article.
-
Adenosine A2A receptor-mediated interactions between Th1+ T cells and the choroid plexus epithelium via IFN-γ signalling control T-Cell infiltration in experimental autoimmune encephalomyelitis.Cell Commun Signal. 2025 Feb 17;23(1):94. doi: 10.1186/s12964-025-02100-7. Cell Commun Signal. 2025. PMID: 39962526 Free PMC article.
-
Choroid plexus-selective inactivation of adenosine A2A receptors protects against T cell infiltration and experimental autoimmune encephalomyelitis.J Neuroinflammation. 2022 Feb 18;19(1):52. doi: 10.1186/s12974-022-02415-z. J Neuroinflammation. 2022. PMID: 35180864 Free PMC article.
-
Involvement of the choroid plexus in central nervous system inflammation.Microsc Res Tech. 2001 Jan 1;52(1):112-29. doi: 10.1002/1097-0029(20010101)52:1<112::AID-JEMT13>3.0.CO;2-5. Microsc Res Tech. 2001. PMID: 11135454 Review.
-
Role of Th17 cells in the pathogenesis of CNS inflammatory demyelination.J Neurol Sci. 2013 Oct 15;333(1-2):76-87. doi: 10.1016/j.jns.2013.03.002. Epub 2013 Apr 8. J Neurol Sci. 2013. PMID: 23578791 Free PMC article. Review.
References
-
- Owens B. Multiple sclerosis. Nature. 2016;540(7631):S1. - PubMed
-
- Marcus R, What. Is Multiple Sclerosis? JAMA. 2022;328(20):2078. - PubMed
-
- Bjornevik K, Munz C, Cohen JI, Ascherio A. Epstein-Barr virus as a leading cause of multiple sclerosis: mechanisms and implications. Nat Rev Neurol. 2023;19(3):160. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

