Overexpression of Wild-Type TMEM43 Improves Cardiac Function in Arrhythmogenic Right Ventricular Cardiomyopathy Type 5
- PMID: 40091736
- PMCID: PMC11984551
- DOI: 10.1161/CIRCRESAHA.124.325848
Overexpression of Wild-Type TMEM43 Improves Cardiac Function in Arrhythmogenic Right Ventricular Cardiomyopathy Type 5
Abstract
Background: Arrhythmogenic right ventricular cardiomyopathy type 5 (ARVC5) is the most aggressive type of ARVC, caused by a fully penetrant missense mutation (p.S358L) in TMEM43 (transmembrane protein 43). Pathologically, the disease is characterized by dilation of the cardiac chambers and fibrofatty replacement of the myocardium, which results in heart failure and sudden cardiac death. Current therapeutic options are limited, and no specific therapies targeting the primary cause of the disease have been proposed.
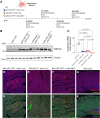
Methods: We investigated whether overexpression of wild-type (WT) TMEM43 could overcome the detrimental effects of the mutant form. We used transgenic mouse models overexpressing either WT or mutant (S358L) TMEM43 to generate a double transgenic mouse line overexpressing both forms of the protein. In addition, we explored if systemic delivery of a codon-optimized self-complementary adeno-associated virus bearing WT-TMEM43 could improve disease progression assessed by ECG and echocardiography.
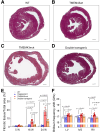
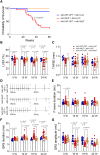
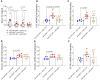
Results: Double transgenic mice overexpressing both WT and mutant TMEM43 forms showed delayed ARVC5 onset, improved cardiac contraction, and reduced ECG abnormalities compared with mice expressing S358L-TMEM43. In addition, cardiomyocyte death and myocardial fibrosis were reduced, with an overall increase in survival. Finally, we demonstrated that a single systemic administration of an adeno-associated virus carrying codon-optimized WT-TMEM43 prevents ventricular dysfunction and ECG abnormalities induced by S358L-TMEM43.
Conclusions: Overexpression of WT-TMEM43 improves the pathological phenotype in a mouse model of ARVC5. Adeno-associated virus-mediated delivery of WT-TMEM43 offers a promising and specific therapy for patients suffering from this highly lethal disease.
Keywords: arrhythmogenic right ventricular dysplasia; cardiomyopathies; mice, transgenic; myocardium.
Conflict of interest statement
The authors would like to acknowledge that this research has led to the filing of a pending patent application. Inventors: B.J. Byrne, M. Corti, E. Lara-Pezzi, L. Lalaguna, and P. García-Pavía. Application number: EP25382006.2. Title: Method for improving cardiac function in arrhythmogenic right ventricular cardiomyopathy type 5. Priority date: 08/01/2025. Applicants: Centro Nacional de Investigaciones Cardiovasculares Carlos III (F.S.P.), Fundación para la Investigación Biomédica del Hospital Universitario Puerta de Hierro Majadahonda, University of Florida Research Foundation, Incorporated.
Figures




Comment in
-
TMEM-ing the Tide: Gene Therapy Holds Promise for ARVC5.Circ Res. 2025 Apr 11;136(8):845-847. doi: 10.1161/CIRCRESAHA.125.326367. Epub 2025 Apr 10. Circ Res. 2025. PMID: 40208928 No abstract available.
References
-
- Corrado D, Basso C, Judge DP. Arrhythmogenic cardiomyopathy. Circ Res. 2017;121:785–802. - PubMed
-
- Arbelo E, Protonotarios A, Gimeno JR, Arbustini E, Barriales-Villa R, Basso C, Bezzina CR, Biagini E, Blom NA, de Boer RA, et al. ; ESC Scientific Document Group. 2023 ESC guidelines for the management of cardiomyopathies: developed by the task force on the management of cardiomyopathies of the European Society of Cardiology (ESC). Eur Heart J. 2023;44:3503–3626. doi: 10.1093/eurheartj/ehad194 - PubMed
-
- Marcus FI, McKenna WJ, Sherrill D, Basso C, Bauce B, Bluemke DA, Calkins H, Corrado D, Cox MGPJ, Daubert JP, et al. . Diagnosis of arrhythmogenic right ventricular cardiomyopathy/dysplasia: proposed modification of the task force criteria. Eur Heart J. 2010;31:806–814. doi: 10.1093/eurheartj/ehq025 - PMC - PubMed
-
- James CA, Jongbloed JDH, Hershberger RE, Morales A, Judge DP, Syrris P, Pilichou K, Domingo AM, Murray B, Cadrin-Tourigny J, et al. . International evidence based reappraisal of genes associated with arrhythmogenic right ventricular cardiomyopathy using the clinical genome resource framework. Circ Genom Precis Med. 2021;14:e003273. doi: 10.1161/CIRCGEN.120.003273 - PMC - PubMed
-
- Merner ND, Hodgkinson KA, Haywood AFM, Connors S, French VM, Drenckhahn JD, Kupprion C, Ramadanova K, Thierfelder L, McKenna W, et al. . Arrhythmogenic right ventricular cardiomyopathy type 5 is a fully penetrant, lethal arrhythmic disorder caused by a missense mutation in the TMEM43 gene. Am J Hum Genet. 2008;82:809–821. doi: 10.1016/j.ajhg.2008.01.010 - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

