Transcriptional insights on the incomplete cytoplasmic maturation and developmental potential of oocytes cultured without granulosa cells in mice
- PMID: 40102748
- PMCID: PMC11921487
- DOI: 10.1186/s12864-025-11455-7
Transcriptional insights on the incomplete cytoplasmic maturation and developmental potential of oocytes cultured without granulosa cells in mice
Erratum in
-
Correction: Transcriptional insights on the incomplete cytoplasmic maturation and developmental potential of oocytes cultured without granulosa cells in mice.BMC Genomics. 2025 Jun 23;26(1):572. doi: 10.1186/s12864-025-11685-9. BMC Genomics. 2025. PMID: 40551118 Free PMC article. No abstract available.
Abstract
Background: Oocyte maturation is crucial for female fertility and embryonic development, encompassing nuclear and cytoplasmic maturation. Supportive cells of follicles, such as granulosa cells, are essential for oocyte growth and maturation. Oocytes can achieve nuclear maturation without granulosa cells during in vitro maturation (IVM). However, there is still a higher chance of incomplete cytoplasmic maturation for these oocytes with mature nuclei compared with oocytes cultured with granulosa cells. Oocytes with incomplete cytoplasmic maturation have lower fertilization rates and developmental potential than mature ones, although underlying mechanisms are poorly understood. Identifying key genes and signaling pathways associated with oocyte cytoplasmic maturation can help further elucidate the maturing process of oocytes and understand the impact of immature oocytes on embryonic development, throwing insights into the strategy to improve the success rate of assisted reproductive technologies.
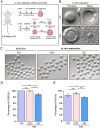
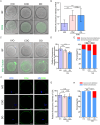
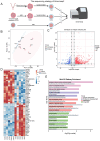
Results: Our study investigated murine oocytes maturing with and without granulosa cells. IVM without granulosa cells yielded oocytes with lower nuclear maturation rates than IVM with granulosa cells and in vivo maturation (IVO). Even though oocytes could achieve nuclear maturation without granulosa cells, they showed incomplete cytoplasmic maturation featuring higher levels of reactive oxygen species, lower mitochondrial density, and higher proportions of cells with abnormal distributions of cortical granules. Of note, oocytes with immature and mature cytoplasm had distinct transcriptional profiles. In the immature oocytes, we observed a deficient mRNA restoration of genes in crucial regulatory pathways of cellular growth and division, potentially affecting embryonic development. Differentially expressed genes (DEGs) between immature and mature oocytes were identified to be highly expressed in different pre-implantation stages, such as the MII oocyte, the 8-cell stage, and the ICM stage. Identified DEGs were enriched in key regulatory pathways of fertilization and embryonic development, such as energy and metabolic pathways. These observations indicated that the impeded development potential of oocytes with immature cytoplasm might be the result of abnormal gene expressions during oocyte maturation.
Conclusions: We show that granulosa cells are important for both nuclear and cytoplasmic maturation of oocytes. Abnormal gene expression in oocytes with incomplete cytoplasmic maturation may be associated with potential defects in fertilization and embryonic development.
Keywords: Embryonic development; Fertilization; Granulosa cells; In vitro maturation; Oocyte cytoplasmic maturation.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Ethics approval and consent to participate: This study is in accordance with ARRIVE guidelines. All procedures executed in mice were approved by the Institutional Animal Care and Use Committee (IACUC) of Peking University Health Science Center Animal Laboratory Center (Beijing, China). Ethical approval for animal survival was given by the Animal Ethics Committee of Peking University Third Hospital (Beijing, China). All mice were sacrificed before the removal of organs based upon the IACUC Guidelines for the euthanasia of experimental animals (Permission No. A2023007). Consent for publication: Not applicable. Competing interests: The authors declare no competing interests.
Figures


Similar articles
-
Biphasic CAPA-IVM Improves Equine Oocyte Quality and Subsequent Embryo Development Without Inducing Genetic Aberrations.Int J Mol Sci. 2025 Jun 8;26(12):5495. doi: 10.3390/ijms26125495. Int J Mol Sci. 2025. PMID: 40564960 Free PMC article.
-
Comparative transcriptome analysis of bovine blastocysts reveals specific effects of the oocyte source and the environments during maturation and early embryo development.BMC Genomics. 2025 Jul 17;26(1):673. doi: 10.1186/s12864-025-11848-8. BMC Genomics. 2025. PMID: 40676537 Free PMC article.
-
Single-cell proteomics analysis of human oocytes during GV-to-MI transition.Hum Reprod. 2025 Jul 1;40(7):1332-1343. doi: 10.1093/humrep/deaf086. Hum Reprod. 2025. PMID: 40359387
-
Intra-cytoplasmic sperm injection versus partial zona dissection, subzonal insemination and conventional techniques for oocyte insemination during in vitro fertilisation.Cochrane Database Syst Rev. 2000;(2):CD001301. doi: 10.1002/14651858.CD001301. Cochrane Database Syst Rev. 2000. Update in: Cochrane Database Syst Rev. 2003;(2):CD001301. doi: 10.1002/14651858.CD001301. PMID: 10796764 Updated.
-
Individualised gonadotropin dose selection using markers of ovarian reserve for women undergoing in vitro fertilisation plus intracytoplasmic sperm injection (IVF/ICSI).Cochrane Database Syst Rev. 2018 Feb 1;2(2):CD012693. doi: 10.1002/14651858.CD012693.pub2. Cochrane Database Syst Rev. 2018. Update in: Cochrane Database Syst Rev. 2024 Jan 4;1:CD012693. doi: 10.1002/14651858.CD012693.pub3. PMID: 29388198 Free PMC article. Updated.
Cited by
-
Correction: Transcriptional insights on the incomplete cytoplasmic maturation and developmental potential of oocytes cultured without granulosa cells in mice.BMC Genomics. 2025 Jun 23;26(1):572. doi: 10.1186/s12864-025-11685-9. BMC Genomics. 2025. PMID: 40551118 Free PMC article. No abstract available.
References
-
- Adhikari D, Liu K. The regulation of maturation promoting factor during prophase I arrest and meiotic entry in mammalian oocytes. Mol Cell Endocrinol. 2014;382(1):480–7. - PubMed
-
- Coticchio G, Dal Canto M, Mignini Renzini M, Guglielmo MC, Brambillasca F, Turchi D, et al. Oocyte maturation: gamete-somatic cells interactions, meiotic resumption, cytoskeletal dynamics and cytoplasmic reorganization. Hum Reprod Update. 2015;21(4):427–54. - PubMed
MeSH terms
Substances
Grants and funding
- 2024M750654/the China Postdoctoral Science Foundation
- 2024YFC2706600/National Key Research and Development Program of China
- 82071612, 82192873, 81971381, 82101714, 81771580, 82271699, 82201825/National Natural Science Foundation of China
- 82225019/National Science Fund for Distinguished Young Scholars
- 20240484604, 20220484073/Beijing Nova Program
LinkOut - more resources
Full Text Sources

