Corey-Fuchs reaction enabled synthesis of natural products: a review
- PMID: 40103970
- PMCID: PMC11912353
- DOI: 10.1039/d5ra00619h
Corey-Fuchs reaction enabled synthesis of natural products: a review
Abstract
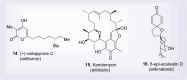
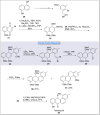
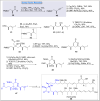
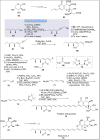
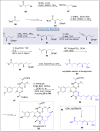
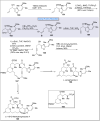
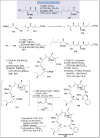
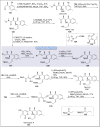
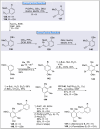
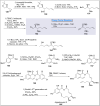
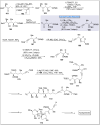
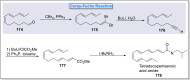
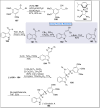
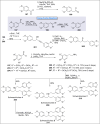
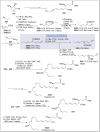
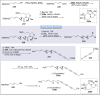
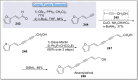
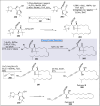
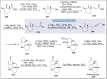
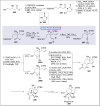
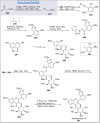
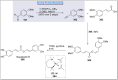
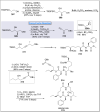
Natural products can be derived from a vast array of animals, plants and microorganisms and are generally characterized by a wide spectrum of bioactive properties such as anti-viral, anti-cancer, anti-inflammatory and anti-bacterial properties. Synthesis of natural products is of paramount importance in various fields including medicine, biotechnology and agriculture. The Corey-Fuchs reaction, also known as the Ramirez Corey-Fuchs reaction, is a pivotal organic transformation and plays a significant role in the synthesis of intricate natural products and their analogues. This review article highlights the development of the Corey-Fuchs reaction in recent years towards the synthesis of complex natural products including polyketides, alkaloids, terpenoids and peptides.
This journal is © The Royal Society of Chemistry.
Conflict of interest statement
There are no conflicts to declare.
Figures








Similar articles
-
The Corey-Seebach Reagent in the 21st Century: A Review.Molecules. 2023 May 26;28(11):4367. doi: 10.3390/molecules28114367. Molecules. 2023. PMID: 37298842 Free PMC article. Review.
-
Novel Bioactive Natural Products from Marine-Derived Penicillium Fungi: A Review (2021-2023).Mar Drugs. 2024 Apr 23;22(5):191. doi: 10.3390/md22050191. Mar Drugs. 2024. PMID: 38786582 Free PMC article. Review.
-
Mukaiyama aldol reaction: an effective asymmetric approach to access chiral natural products and their derivatives/analogues.RSC Adv. 2023 Nov 8;13(47):32975-33027. doi: 10.1039/d3ra05058k. eCollection 2023 Nov 7. RSC Adv. 2023. PMID: 38025859 Free PMC article. Review.
-
Electrochemically modified Corey-Fuchs reaction for the synthesis of arylalkynes. The case of 2-(2,2-dibromovinyl)naphthalene.Beilstein J Org Chem. 2018 Apr 23;14:891-899. doi: 10.3762/bjoc.14.76. eCollection 2018. Beilstein J Org Chem. 2018. PMID: 29765470 Free PMC article.
-
A Case for Bond-Network Analysis in the Synthesis of Bridged Polycyclic Complex Molecules: Hetidine and Hetisine Diterpenoid Alkaloids.Angew Chem Int Ed Engl. 2020 Jun 26;59(27):10722-10731. doi: 10.1002/anie.201909656. Epub 2020 Apr 15. Angew Chem Int Ed Engl. 2020. PMID: 31808282 Free PMC article. Review.
References
Publication types
LinkOut - more resources
Full Text Sources

