Emerging clinical and research approaches in targeted therapies for high-risk neuroblastoma
- PMID: 40104501
- PMCID: PMC11913827
- DOI: 10.3389/fonc.2025.1553511
Emerging clinical and research approaches in targeted therapies for high-risk neuroblastoma
Abstract
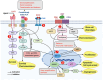
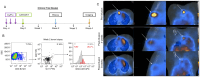
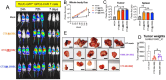
Neuroblastoma is a pediatric cancer that originates from neural crest cells and is the most common extracranial solid tumor in children under five years of age. While low-risk neuroblastoma often regresses spontaneously, high-risk neuroblastoma poses a significant clinical challenge. Recent advances in understanding neuroblastoma's molecular mechanisms have led to the development of targeted therapies that aim to selectively inhibit specific pathways involved in tumor growth and progression, improving patient outcomes while minimizing side effects. This review provides a comprehensive review of neuroblastoma biology and emerging therapeutic strategies. Key topics include (a) immunotherapies and immunotargets, (b) non-coding RNAs (long non-coding RNA, microRNA, and circular RNA), (c) molecular biomarkers and pathways, and (d) limitations and future directions.
Keywords: clinical and preclinical research; high-risk neuroblastoma; neuroblastoma biology; pediatric cancer; targeted therapy.
Copyright © 2025 AlKhazal, Chohan, Ross, Kim and Brown.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


References
-
- Anderson D, Carnahan J, Michelsohn A, Patterson P. Antibody markers identify a common progenitor to sympathetic neurons and chromaffin cells in vivo and reveal the timing of commitment to neuronal differentiation in the sympathoadrenal lineage. J Neurosci. (1991) 11:3507–19. doi: 10.1523/JNEUROSCI.11-11-03507.1991 - DOI - PMC - PubMed

