MRI assessment of body composition for prediction of therapeutic response to biologic agents in patients with Crohn's disease
- PMID: 40106118
- PMCID: PMC11923306
- DOI: 10.1186/s13244-025-01930-w
MRI assessment of body composition for prediction of therapeutic response to biologic agents in patients with Crohn's disease
Abstract
Objectives: Altered body fat and muscle mass in Crohn's disease (CD) have been linked to adverse disease course and outcomes. Prediction of treatment response or remission (RoR) of small bowel CD (SBCD) to biologic therapy remains challenging. We aimed to establish the prognostic value of body composition parameters measured using MR enterography (MRE) for RoR at 1 year in patients with SBCD commencing biologic therapy.
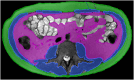
Methods: Participants were identified from those recruited to a prospective, multicentre study investigating the predictive ability of motility MRI for 1 year RoR in patients starting biologic therapy for active SBCD (MOTILITY trial). Myopenia, skeletal muscle:fat and visceral:subcutaneous fat were measured from baseline MRE. RoR at 1 year was judged using a composite of clinical and morphological MRE parameters. We compared the likelihood of RoR in patients with and without myopenia or low skeletal muscle:fat using logistic regression models.
Results: Ninety-six participants were included (mean age 38.2 years; 40 (42%) female). There were 34 (35%) responders. There was no significant difference in RoR at 1 year between those patients with and without skeletal muscle myopenia (OR: 0.85, 95% CI: 0.27, 2.66, p-value: 0.78), or those with or without low skeletal muscle:fat (OR: 0.71, 95% CI: 0.19, 2.71, p-value: 0.62).
Conclusions: Body composition parameters demonstrated no value for predicting therapeutic RoR in patients commencing biologic therapy for SBCD.
Critical relevance statement: Prediction of response to biologic therapy in small bowel Crohn's disease (SBCD) remains challenging. Body composition parameters cannot predict biologic therapeutic response or remission for SBCD reliably.
Key points: Altered body fat and muscle mass in Crohn's disease have been linked to adverse outcomes. Prediction of response to biologic therapy in small bowel CD (SBCD) would be useful for treatment optimisation. Body composition parameters measured using MRI cannot reliably predict biological therapeutic response or remission for SBCD.
Keywords: Biological therapy; Body composition; Crohn’s disease; Magnetic resonance imaging.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Ethics approval and consent to participate: This was a prespecified substudy of the MOTILITY trial (ISRCTN14481560). The study was ethically approved by the NHS West Midlands Research Ethics Committee: 17/WM/0106. All participants gave written informed consent. Consent for publication: Not applicable. Competing interests: G.W.M. is in receipt of research funding from AstraZeneca, Bristol Myers Squibb, Jansen, Alimentiv and Pfizer; provided consultancy services on advisory boards for Jansen, Pfizer, Abbvie; is a consultant for Alimetiv Inc. and Satisfai health. S.T. has received grants/research support from AbbVie, Celgene, Celsius, ECCO, Galapagos, Helmsley Trust, IOIBD, Janssen, Lilly, Pfizer, Takeda, UKIERI, Vifor, and Norman Collisson Foundation; consulting Fees from Alimentiv, Apexian, Apollo, Arcturis, Arena, AstraZeneca, BMS, Buhlmann, Celgene, ChemoCentryx, Clario, Cosmo, Dynavax, Endpoint Health, EQrX, Equillium, Ferring, Galapagos, Genentech/Roche, Gilead, GSK, Janssen, Lilly, Mestag, Microbiotica, ONO, Pfizer, Protagonist, Sanofi, Satisfai, Sensyne Health, Sorriso, Syndermix, Takeda, Theravance, Topivert, Tr1X Bio, UCB Pharma, Vifor; speaker fees from BMS, Ferring, Janssen, Lilly, Pfizer, Sun Pharma, Takeda and has share options in Satisfai Health Inc. H.L. has received speaking honoraria from Takeda Pharmaceuticals. G.B. is a consultant for Alimentiv, is an employee of Motilent, has share options in Motilent and owns patent P295276.US.02, system to characterise topology and morphology of fistulae from medical imaging data. A.M. is CEO of Motilent. S.A.T. receives research support from Takeda, is a consultant to AstraZeneca and shareholder in Motilent. The remaining authors declare that they have no competing interests.
Figures

References
-
- Burnham JM, Shults J, Semeao E et al (2005) Body-composition alterations consistent with cachexia in children and young adults with Crohn disease. Am J Clin Nutr 82:413–420 - PubMed
-
- Vaisman N, Dotan I, Halack A, Niv E (2006) Malabsorption is a major contributor to underweight in Crohn’s disease patients in remission. Nutrition 22:855–859 - PubMed
-
- Valentini L, Schaper L, Buning C et al (2008) Malnutrition and impaired muscle strength in patients with Crohn’s disease and ulcerative colitis in remission. Nutrition 24:694–702 - PubMed
-
- Schneider S, Al-Jaouni R, Filippi J et al (2008) Sarcopenia is prevalent in patients with Crohn’s disease in clinical remission. Inflamm Bowel Dis 14:1562–1568 - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources

