Low-intensity pulsed ultrasound elevates blood pressure for shock
- PMID: 40106546
- PMCID: PMC11922025
- DOI: 10.1126/sciadv.ads6947
Low-intensity pulsed ultrasound elevates blood pressure for shock
Abstract
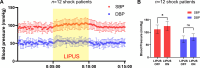
Fluid replacement is the primary treatment for life-threatening shock but is challenging in harsh environments. This study explores low-intensity pulsed ultrasound (LIPUS) as a resuscitation strategy. Cervical LIPUS stimulation effectively elevated blood pressure in shocked rats. It also improved cerebral and multiorgan perfusion. Mechanistically, LIPUS activated pathways related to sympathetic nerve excitation and vascular smooth muscle contraction, increasing plasma catecholamines and stimulating blood pressure-regulating neural nuclei. Partial sympathetic nerve transection reduced LIPUS efficacy, while complete inhibition of these nuclei abolished the response. Preliminary clinical trials demonstrated LIPUS's ability to raise blood pressure in shock patients. The findings suggest that LIPUS enhances sympathetic nerve activity and activates blood pressure-regulating nuclei, offering a noninvasive, neuromodulation-based approach to shock treatment. This method holds potential for improving blood pressure and organ perfusion in shock patients, especially in resource-limited environments.
Figures



References
-
- Kauvar D. S., Lefering R., Wade C. E., Impact of hemorrhage on trauma outcome: An overview of epidemiology, clinical presentations, and therapeutic considerations. J. Trauma 60, S3–S11 (2006). - PubMed
-
- J. E. Tintinalli, J. S. Stapczynski, O. J. Ma, D. M. Yealy, G. D. Meckler, D. M. Cline, Tintinalli's Emergency Medicine: A Comprehensive Study Guide, 8e (McGraw Hill Education, 2016).
-
- Cannon J., Morrison J., Lauer C., Grabo D., Polk T., Blackbourne L., Dubose J., Rasmussen T., Resuscitative endovascular balloon occlusion of the aorta (REBOA) for hemorrhagic shock. Mil. Med. 183, 55–59 (2018). - PubMed
-
- Angus D. C., van der Poll T., Severe sepsis and septic shock. N. Engl. J. Med. 369, 840–851 (2013). - PubMed
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

