USP8 and Hsp70 regulate endoreplication by synergistically promoting Fzr deubiquitination and stabilization
- PMID: 40106570
- PMCID: PMC11922063
- DOI: 10.1126/sciadv.adq9111
USP8 and Hsp70 regulate endoreplication by synergistically promoting Fzr deubiquitination and stabilization
Abstract
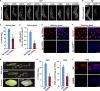
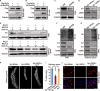
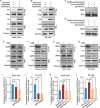
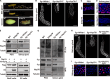
Endoreplication is characterized by multiple rounds of DNA replication without cell division and determines the growth and final size of endoreplicating cells and tissues in eukaryotes. The cyclic ubiquitination and degradation of several cell cycle regulators are required for endoreplication progression. However, the deubiquitinase that deubiquitinates and stabilizes key factors to modulate endoreplication remains unknown. Here, we found in the endoreplicating Drosophila salivary gland and Bombyx silk gland that the depletion of ubiquitin-specific peptidase 8 (USP8) led to endoreplication arrest and a decrease in gland size. Mechanistically, we showed that USP8 interacted with the Fizzy-related (Fzr) protein, a conserved master regulator of endoreplication, thereby deubiquitinating and stabilizing Fzr to modulate endoreplication. Moreover, the molecular chaperone heat shock protein 70 (Hsp70) mediated proper folding of Fzr and increased the interaction between Fzr and USP8, thereby promoting the deubiquitination and stabilization of Fzr. Together, our study demonstrates that USP8 and Hsp70 regulate endoreplication by synergistically maintaining Fzr stability though deubiquitination.
Figures


Similar articles
-
A novel transcriptional cascade is involved in Fzr-mediated endoreplication.Nucleic Acids Res. 2020 May 7;48(8):4214-4229. doi: 10.1093/nar/gkaa158. Nucleic Acids Res. 2020. PMID: 32182338 Free PMC article.
-
The ubiquitin-specific protease USP8 deubiquitinates and stabilizes Cx43.J Biol Chem. 2018 May 25;293(21):8275-8284. doi: 10.1074/jbc.RA117.001315. Epub 2018 Apr 6. J Biol Chem. 2018. PMID: 29626091 Free PMC article.
-
The Endosome-associated Deubiquitinating Enzyme USP8 Regulates BACE1 Enzyme Ubiquitination and Degradation.J Biol Chem. 2016 Jul 22;291(30):15753-66. doi: 10.1074/jbc.M116.718023. Epub 2016 Jun 14. J Biol Chem. 2016. PMID: 27302062 Free PMC article.
-
The oncogenic role of ubiquitin specific peptidase (USP8) and its signaling pathways targeting for cancer therapeutics.Arch Biochem Biophys. 2021 Apr 15;701:108811. doi: 10.1016/j.abb.2021.108811. Epub 2021 Feb 16. Arch Biochem Biophys. 2021. PMID: 33600786 Review.
-
Ubiquitin-specific protease 8 (USP8/UBPy): a prototypic multidomain deubiquitinating enzyme with pleiotropic functions.Biochem Soc Trans. 2019 Dec 20;47(6):1867-1879. doi: 10.1042/BST20190527. Biochem Soc Trans. 2019. PMID: 31845722 Free PMC article. Review.
Cited by
-
The Rheb-mTORC1 Coordinates Cell Cycle Progression and Endoreplication in Bombyx mori.Insects. 2025 Jun 20;16(7):647. doi: 10.3390/insects16070647. Insects. 2025. PMID: 40725279 Free PMC article.
References
-
- Edgar B. A., Zielke N., Gutierrez C., Endocycles: A recurrent evolutionary innovation for post-mitotic cell growth. Nat. Rev. Mol. Cell Biol. 15, 197–210 (2014). - PubMed
-
- Breuer C., Ishida T., Sugimoto K., Developmental control of endocycles and cell growth in plants. Curr. Opin. Plant Biol. 13, 654–660 (2010). - PubMed
-
- Patra S., Naik P. P., Mahapatra K. K., Alotaibi M. R., Patil S., Patro B. S., Sethi G., Efferth T., Bhutia S. K., Recent advancement of autophagy in polyploid giant cancer cells and its interconnection with senescence and stemness for therapeutic opportunities. Cancer Lett. 590, 216843 (2024). - PubMed
-
- Dhawan S., Gopinathan K. P., Cell cycle events during the development of the silk glands in the mulberry silkworm Bombyx mori. Dev. Genes Evol. 213, 435–444 (2003). - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

