Potential of High-Intensity Focused Ultrasound in Enamel Remineralization
- PMID: 40108508
- PMCID: PMC12209549
- DOI: 10.1177/00220345251323869
Potential of High-Intensity Focused Ultrasound in Enamel Remineralization
Abstract
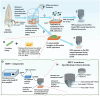
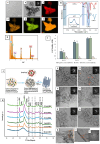
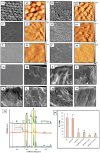
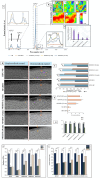
Remineralization is an essential interventional strategy for intercepting enamel white spot lesions (WSLs). Given the limitations of both natural and/or fluoride-mediated repair processes, there is a need to develop novel strategies for repairing enamel WSLs via a minimally invasive approach while restoring the unique ultrastructural integrity and functional properties. Inspired by the unique capability of high-intensity focused ultrasound (HIFU) in facilitating the crystallization process, we propose a novel strategy of employing HIFU for in vitro repair of WSLs through synergizing the crystallization process required for hydroxyapatite (HAP) formation from its precursor (calcium phosphate ion clusters; CPICs). Following CPIC formulation and characterization including the resultant amorphous calcium phosphate (ACP), the effect of HIFU on the ACP-to-HAP transition on the amorphous substrate was investigated using transmission electron microscopy and high-resolution transmission electron microscopy, selected area electron diffraction, and X-ray diffraction (XRD). The results showed profound amorphous-to-crystalline phase transition, within 5- to 30-min HIFU exposure, whereas the long axis of the resultant HAP corresponded with the (002) plane, and a lattice spacing of 0.34 nm indicated a preferred c-axis growth direction consistent with the orientation of natural enamel crystallites. For enamel repair, artificial WSLs were created on enamel specimens and then subjected to CPICs, followed by HIFU exposure for 2.5, 5, or 10 min. Scanning electron and atomic force microscopies revealed the decreased surface roughness and the gradual obliteration in the WSL porous structure with continuous linear coaxial arrangement of HAP crystallites filling the prismatic/interprismatic gaps closely resembling sound enamel specifically with 5-min HIFU exposure. Enamel WSL ultrastructural repair was further confirmed from XRD and Raman spectral analyses with the associated regaining of mineral density and nanomechanical properties as reflected from micro-computed tomography (CT) and nanoindentation results, respectively. Micro-CT further validated the subsurface remineralization of WSLs with HIFU exposure. Within the same exposure parameters, HIFU exhibited a potent antibiofilm effect against Streptococcus mutans. This study introduced a new approach for remineralizing enamel WSLs through the potent synergy between HIFU and CPICs.
Keywords: biomaterials; cariogenic biofilms; enamel repair; enamel white spot lesions; minimal invasive dentistry; ultrasonic energy.
Conflict of interest statement
Declaration of Conflicting InterestsThe authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Figures

Similar articles
-
Evaluation of enamel remineralization potential and anticariogenic efficacy of polydopamine coated biogenic amorphous calcium phosphate.Clin Oral Investig. 2025 May 20;29(6):302. doi: 10.1007/s00784-025-06384-4. Clin Oral Investig. 2025. PMID: 40389610
-
The Synergistic Effect of High Intensity Focused Ultrasound on In-vitro Remineralization of Tooth Enamel by Calcium Phosphate Ion Clusters.Int J Nanomedicine. 2024 Jun 6;19:5365-5380. doi: 10.2147/IJN.S464998. eCollection 2024. Int J Nanomedicine. 2024. PMID: 38859951 Free PMC article.
-
Development of a novel Agarose/Nano-Hydroxyapatite/Grape seed extract hydrogel for biomimetic remineralization of demineralized human enamel (An In-Vitro Study).Sci Rep. 2025 Jul 18;15(1):26086. doi: 10.1038/s41598-025-11490-0. Sci Rep. 2025. PMID: 40681738 Free PMC article.
-
The effect of nano-hydroxyapatite on white spot lesions: A systematic review and meta-analysis.J Dent. 2024 Dec;151:105402. doi: 10.1016/j.jdent.2024.105402. Epub 2024 Oct 10. J Dent. 2024. PMID: 39395626
-
Long term remineralizing effect of casein phosphopeptide amorphous calcium phosphate in white spot lesions: A systematic review and meta-analysis.Technol Health Care. 2024;32(3):1239-1254. doi: 10.3233/THC-230722. Technol Health Care. 2024. PMID: 37599550
References
-
- Aati S, Shrestha B, Fawzy A. 2022. Cytotoxicity and antimicrobial efficiency of ZrO2 nanoparticles reinforced 3D printed resins. Dent Mater. 38(8):1432–1442. - PubMed
-
- Bang JH, Suslick KS. 2010. Applications of ultrasound to the synthesis of nanostructured materials. Adv Mater. 22(10):1039–1059. - PubMed
-
- Bubnowicz L, França R. 2022. Calcium and fluoride interaction in human enamel at nanoscale: an XPS assessment. Appl Surf Sci Adv. 11:100306. doi: 10.1016/j.apsadv.2022.100306 - DOI
-
- Carden A, Morris MD. 2000. Application of vibrational spectroscopy to the study of mineralized tissues (review). J Biomed Opt. 5(3):259–268. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

