Accuracy of plasma biomarkers to detect Alzheimer's disease proteinopathy prior to dementia
- PMID: 40108829
- PMCID: PMC11922805
- DOI: 10.1002/alz.14570
Accuracy of plasma biomarkers to detect Alzheimer's disease proteinopathy prior to dementia
Abstract
Introduction: Plasma biomarkers sensitive to Alzheimer's disease (AD) proteinopathy prior to the onset of dementia have significant implications for early detection.
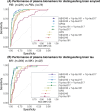
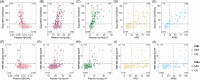
Methods: In 304 individuals without dementia, we investigated whether C2N Diagnostics' mass spectrometry (MS)-based plasma biomarkers (amyloid beta 42/40, %phosphorylated tau [p-tau]181, and %p-tau217) and amyloid probability scores (APS, PrecivityAD and APS2, PrecivityAD2) are associated with brain amyloid, brain tau, or preclinical cognitive decline.
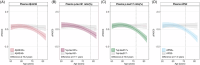
Results: In this cohort study, %p-tau217 and the APS2 had high discriminative accuracy (area under the curve > 0.93) for identifying elevated brain amyloid and tau and were associated with faster preclinical cognitive decline. Using %p-tau217 or the APS2 in a theoretical AD trial screening scenario reduced amyloid positron emission tomography imaging costs up to 41% or 45%, respectively.
Discussion: These findings suggest that C2N Diagnostics' MS-based plasma biomarkers can detect brain amyloid and tau with high accuracy prior to dementia and could aid in identifying candidates for clinical trials or therapeutic intervention.
Highlights: C2N plasma biomarkers differentiated Alzheimer's disease proteinopathy status prior to dementia. Plasma %phosphorylated tau (p-tau)217 and the C2N Diagnostics PrecivityAD2 (APS2) were concordant with amyloid and tau positron emission tomography status. Plasma %p-tau217 and the APS2 were associated with preclinical cognitive decline.
Keywords: biomarkers; mass spectrometry; plasma; positron emission tomography; preclinical Alzheimer's disease.
© 2025 The Author(s). Alzheimer's & Dementia published by Wiley Periodicals LLC on behalf of Alzheimer's Association.
Conflict of interest statement
Dr. Johnson has served as an advisor to Roche Diagnostics, AlzPath, and Enigma Biosciences and has received research support (sponsoring of an unrelated observational study) from Cerveau Technologies. Drs. Kirmess, Meyer, Yarasheski, West, Verghese, and Braunstein are paid employees of C2N Diagnostics. No other disclosures were reported. Author disclosures are available in the supporting information.
Figures
References
-
- Budd Haeberlein S, Aisen PS, Barkhof F, et al. Two randomized phase 3 studies of aducanumab in early Alzheimer's disease. J Prev Alzheimer Dis. 2022;9(2):197‐210. - PubMed
-
- van Dyck CH, Swanson CJ, Aisen P, et al. Lecanemab in early Alzheimer's disease. N Engl J Med. 2022;388(1):9‐21. - PubMed
-
- Blennow K, Mattsson N, Scholl M, Hansson O, Zetterberg H. Amyloid biomarkers in Alzheimer's disease. Trends Pharmacol Sci. 2015;36(5):297‐309. - PubMed
-
- Ossenkoppele R, van der Kant R, Hansson O. Tau biomarkers in Alzheimer's disease: towards implementation in clinical practice and trials. Lancet Neurol. 2022;21(8):726‐734. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

