Epigenetic regulators combined with tumour immunotherapy: current status and perspectives
- PMID: 40119465
- PMCID: PMC11929245
- DOI: 10.1186/s13148-025-01856-6
Epigenetic regulators combined with tumour immunotherapy: current status and perspectives
Abstract
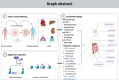
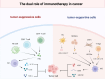
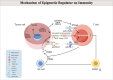
Immunotherapy, particularly immune checkpoint inhibitor therapy, has demonstrated clinical benefits in solid tumours. Despite its satisfactory clinical efficacy, it still faces several issues, such as limited eligibility, low response rates and cytotoxicity. Cancer epigenetics implies that tumour cells exhibit unique phenotypes because of their unique characteristics, thus reprogramming of the epigenome holds promise for cancer therapy. Epigenetic regulation plays an important role in regulating gene expression during tumour development and maintenance. Epigenetic regulators induce cancer cell cycle arrest, apoptosis and differentiation of cancer cells, thereby exerting anti-tumour effects. Recent studies have revealed a significant correlation between epigenetic regulatory factors and immune checkpoint therapy. Epigenetics can modulate various aspects of the tumour immune microenvironment and immune response to enhance the sensitivity of immunotherapy, such as lowering the concentration required and mitigating cytotoxicity. This review primarily discusses DNA methyltransferase inhibitors, histone deacetylase inhibitors, enhancer of zeste homolog 2 inhibitors and lysine-specific demethylase 1 inhibitors, which are associated with transcriptional repression. This repression alters the expression of genes involved in the immune checkpoint, thereby enhancing the effectiveness of immunotherapy. We also discuss the potential and challenges of tumour immunotherapy and highlight its advantages, application challenges and clinical research on integrating epigenetic regulatory factors with tumour immunotherapy.
Keywords: DNA methyltransferase inhibitor; Epigenetic regulation; Histone deacetylase inhibitors; Immune check inhibitor; Tumour immunotherapy.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Consent for publication: The authors declare no competing interests. Competing interests: The authors declare no competing interests.
Figures


Similar articles
-
Epigenetic Mechanisms of Resistance to Immune Checkpoint Inhibitors.Biomolecules. 2020 Jul 16;10(7):1061. doi: 10.3390/biom10071061. Biomolecules. 2020. PMID: 32708698 Free PMC article. Review.
-
Epigenetics and immunotherapy: The current state of play.Mol Immunol. 2017 Jul;87:227-239. doi: 10.1016/j.molimm.2017.04.012. Epub 2017 May 14. Mol Immunol. 2017. PMID: 28511092 Review.
-
Impaired ARID1A expression attenuated the immune response in gastric cancer via histone acetylation.Clin Epigenetics. 2025 Jan 3;17(1):2. doi: 10.1186/s13148-024-01805-9. Clin Epigenetics. 2025. PMID: 39754248 Free PMC article.
-
Epigenetic modulation in cancer immunotherapy.Curr Opin Pharmacol. 2017 Aug;35:48-56. doi: 10.1016/j.coph.2017.05.006. Epub 2017 Jun 10. Curr Opin Pharmacol. 2017. PMID: 28609681 Review.
-
Pharmacological targeting of the cancer epigenome.Nat Cancer. 2024 Jun;5(6):844-865. doi: 10.1038/s43018-024-00777-2. Epub 2024 Jun 27. Nat Cancer. 2024. PMID: 38937652 Free PMC article. Review.
Cited by
-
Comorbidity of hypertension and lung cancer: interplay of genetics and environment.Discov Oncol. 2025 Aug 13;16(1):1548. doi: 10.1007/s12672-025-03323-3. Discov Oncol. 2025. PMID: 40804226 Free PMC article. Review.
-
Immunosenescence and cancer: molecular hallmarks, tumor microenvironment remodeling, and age-specific immunotherapy challenges.J Hematol Oncol. 2025 Aug 22;18(1):81. doi: 10.1186/s13045-025-01735-w. J Hematol Oncol. 2025. PMID: 40846970 Free PMC article. Review.
-
Advances in the regulation of macrophage polarization by the tumor microenvironment.Discov Oncol. 2025 Aug 6;16(1):1487. doi: 10.1007/s12672-025-03258-9. Discov Oncol. 2025. PMID: 40770163 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

