Assessing the impact of a self-guided digital intervention for fear of cancer recurrence (iConquerFear) in ovarian cancer survivors: a pilot randomised waitlist-controlled trial
- PMID: 40122804
- PMCID: PMC11929991
- DOI: 10.1186/s12885-025-13639-6
Assessing the impact of a self-guided digital intervention for fear of cancer recurrence (iConquerFear) in ovarian cancer survivors: a pilot randomised waitlist-controlled trial
Abstract
Background: Approximately 50% of ovarian cancer (OC) survivors report fear of cancer recurrence/progression (FCR/P) as the most challenging aspect of living with cancer. This pilot, randomised waitlist-controlled trial aimed to evaluate the feasibility, acceptability, and safety of iConquerFear, a self-guided online FCR intervention for OC survivors.
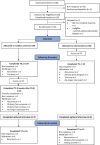
Methods: Stage I-III OC survivors were recruited via Ovarian Cancer Australia (OCA) between October-December 2022. Participants were randomised to access iConquerFear immediately (intervention) or after 8 weeks (waitlist-control). Primary outcomes were feasibility, acceptability, and safety. Secondary outcomes included: engagement barriers/enablers, perceived impact of iConquerFear, and suggested improvements via semi-structured interviews. Exploratory outcomes included group differences in FCR and FoP after iConquerFear use.
Results: Of 62 eligible survivors, 55 (61%) were randomised (intervention n = 29; control n = 26). At baseline 55% (30/55) reported severe FCR (FCRI-SF ≥ 22). Of those randomised, 51% (n = 28) accessed iConquerFear; 16/28 (57%) users completed ≥ 3/5 modules. Mean post-intervention acceptability score (IEUQ) was 3/4 (SD = 0.8). Three (11%) users withdrew due to distress from iConquerFear. Qualitative interviews (n = 13) identified 6 key themes (e.g., participant factors influencing engagement). Differences between intervention and control group changes in FCR/P were non-significant.
Conclusions: iConquerFear does not appear appropriate for OC survivors in its current format due to limited engagement, varied acceptability, safety concerns and minimal group differences in FCR/P after iConquerFear use. More work is needed regarding how to augment online interventions addressing sensitive issues such as FCR/P in OC survivors (e.g., offering complementary in-person support) to ensure feasibility, acceptability and safety.
Trial registration: This trial is registered with ANZCTR.org (ACTRN12622000592741p) on 21 April 2022.
Keywords: Fear of cancer recurrence; Fear of progression; Online; Ovarian cancer; Self management; Web-based; eHealth.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Ethics approval and consent to participate: Ethical approval was obtained from the University of New South Wales Ethics Committee (approval number: HC220186). Informed consent was obtained from all participants. Consent for publication: Not applicable. Competing interests: Allan ‘Ben’ Smith, Adeola Bamgboje-Ayodele, Lisa Beatty, Alison Pearce, Joanna Fardell, and Afaf Girgis hold a copyright licensed to Blue Note Therapeutics, Inc., for the ConquerFear and iConquerFear interventions. All other authors have no competing interests to declare. Additional files: Additional file 1: Semi-structured interview schedule (Additional File 1.docx). Additional file 2: Measures for exploratory outcomes (Additional File 2.docx).
Figures
References
-
- Cabasag CJ, Fagan PJ, Ferlay J, Vignat J, Laversanne M, Liu L, van der Aa MA, Bray F, Soerjomataram I. Ovarian cancer today and tomorrow: a global assessment by world region and Human Development Index using GLOBOCAN 2020. Int J Cancer. 2022;151(9):1535–41. - PubMed
-
- Ovarian Cancer Statistics in Australia. [https://www.canceraustralia.gov.au/cancer-types/ovarian-cancer/statistics]
-
- Lisy K, Langdon L, Piper A, Jefford M. Identifying the most prevalent unmet needs of cancer survivors in Australia: a systematic review. Asia-Pac J Clin Oncol. 2019;15(5):e68–78. - PubMed
-
- Miroševič Š, Prins JB, Selič P, Zaletel Kragelj L, Klemenc Ketiš Z. Prevalence and factors associated with unmet needs in post-treatment cancer survivors: a systematic review. Eur J Cancer Care. 2019;28(3):e13060. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous