PRMT1-mediated modification of H4R3me2a promotes liver cancer progression by enhancing the transcriptional activity of SOX18
- PMID: 40130992
- PMCID: PMC11936655
- DOI: 10.1097/HC9.0000000000000647
PRMT1-mediated modification of H4R3me2a promotes liver cancer progression by enhancing the transcriptional activity of SOX18
Abstract
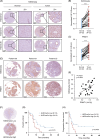
Background: HCC is one of the most prevalent and deadliest malignancies worldwide, with a poor prognosis. Altered histone modifications have been shown to play a significant role in HCC. However, the biological roles and clinical relevance of specific histone modifications, such as the asymmetric dimethylation on arginine 3 of histone H4 (H4R3me2a), remain poorly understood in HCC.
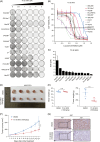
Methods: In this study, immunohistochemical staining was performed to assess histone H4R3me2a modification in 32 pairs of HCC tissues and corresponding adjacent nontumor liver tissues. Cellular-level experiments and subcutaneous xenograft models in nude mice were used to investigate the effects of silencing protein arginine methyltransferase 1 (PRMT1) with shRNA or pharmacologically blocking PRMT1 activity on HCC cell proliferation, migration, and invasion. RNA-seq analysis combined with Chip-qPCR validation was employed to explore the regulatory mechanism of PRMT1 on SOX18 expression. The downstream target of SOX18 was identified using the JASPAR database and a dual-luciferase reporter system.
Results: The level of histone H4R3me2a modification was significantly elevated in HCC tissues and closely associated with poor prognosis in patients with HCC. Silencing PRMT1 or pharmacologically inhibiting its activity effectively suppressed the proliferation, migration, and invasion of HCC cells. Mechanistically, PRMT1 was found to regulate SOX18 expression by modulating histone H4R3me2a modification in the SOX18 promoter region. LOXL1 was identified as a downstream target of the transcription factor SOX18.
Conclusions: This study revealed the clinical relevance of histone H4R3me2a modification in HCC and demonstrated that PRMT1 promotes malignant behavior in HCC cells by modulating H4R3me2a modification in the SOX18 promoter region. The findings elucidate the role and molecular mechanism of PRMT1-mediated histone H4R3me2a modification in HCC progression and highlight the potential clinical applications of PRMT1 inhibitors. These results may provide new insights into the treatment of HCC.
Copyright © 2025 The Author(s). Published by Wolters Kluwer Health, Inc. on behalf of the American Association for the Study of Liver Diseases.
Conflict of interest statement
The authors have no conflicts to report.
Figures



References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

