Bacterial vaginosis associates with dysfunctional T cells and altered soluble immune factors in the cervicovaginal tract
- PMID: 40131862
- PMCID: PMC12077898
- DOI: 10.1172/JCI184609
Bacterial vaginosis associates with dysfunctional T cells and altered soluble immune factors in the cervicovaginal tract
Abstract
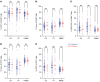
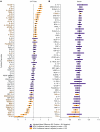
BACKGROUNDBacterial vaginosis (BV) is a dysbiosis of the vaginal microbiome that is prevalent among reproductive-age females worldwide. Adverse health outcomes associated with BV include an increased risk of sexually acquired HIV, yet the immunological mechanisms underlying this association are not well understood.METHODSTo investigate BV-driven changes to cervicovaginal tract (CVT) and circulating T cell phenotypes, Kinga Study participants with or without BV provided vaginal tract (VT) and ectocervical (CX) tissue biopsies and PBMC samples.RESULTSHigh-parameter flow cytometry revealed an increased frequency of cervical CD4+ conventional T (Tconv) cells expressing CCR5 in BR+ versus BR- women. However, we found no difference in the number of CD3+CD4+CCR5+ cells in the CX or VT of BV+ versus BV- individuals, suggesting that BV-driven increased HIV susceptibility may not be solely attributed to increased CVT HIV target cell abundance. Flow cytometry also revealed that individuals with BV had an increased frequency of dysfunctional CX and VT CD39+ Tconv and CX tissue-resident CD69+CD103+ Tconv cells, reported to be implicated in HIV acquisition risk and replication. Many soluble immune factor differences in the CVT further support that BV elicits diverse and complex CVT immune alterations.CONCLUSIONOur comprehensive analysis expands on potential immunological mechanisms that may underlie the adverse health outcomes associated with BV, including increased HIV susceptibility.TRIAL REGISTRATIONClinicalTrials.gov NCT03701802.FUNDINGThis work was supported by National Institutes of Health grants R01AI131914, R01AI141435, and R01AI129715.
Keywords: AIDS/HIV; Immunology; Infectious disease; T cells.
Figures