A novel in-vitro expression assay by LC/MS/MS enables multi-antigen mRNA vaccine characterization
- PMID: 40133349
- PMCID: PMC11937333
- DOI: 10.1038/s41598-025-94616-8
A novel in-vitro expression assay by LC/MS/MS enables multi-antigen mRNA vaccine characterization
Abstract
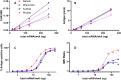
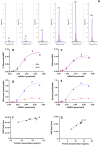
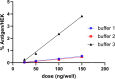
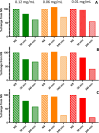
The new era of messenger RNA (mRNA) vaccines has led to development of a novel, state-of-the-art characterization method for this class of molecules. Currently, flow cytometry-based assays with antigen-specific antibodies are utilized for monitoring in-vitro expression (IVE) of mRNA. Here we present development, optimization, and application of an in-vitro expression liquid chromatography tandem mass spectrometry (IVE-LC/MS/MS) assay as an orthogonal method to IVE-flow cytometry that can be used for in-depth characterization of the expressed protein antigens and monitoring their relative expression levels in the cell post-mRNA transfection. The IVE-LC/MS/MS assessment accomplished the detection of influenza hemagglutinin (HA) antigens of four distinct strains simultaneously. The workflow is presented here, highlighting the optimization of all necessary steps required for protein purification and mass spectrometry method setup. The IVE-LC/MS/MS assay is a robust and versatile technique that complements the IVE-flow cytometry method and offers several advantages, such as being antibody-free, capable of multiplexing, and highly sensitive and selective. The various studies in this work, including evaluating dose-response relationships, refining transfection protocols, and examining mRNA-LNP stability under various conditions showcase the significant benefits of applying IVE-LC/MS/MS across different experimental settings. IVE-LC/MS/MS is a powerful tool for understanding and improving the performance and quality of mRNA LNPs.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Competing interests: All authors are employees of Pfizer Inc. Some authors may be shareholders of Pfizer Inc. The authors declare no other competing interests.
Figures




References
-
- Zhong, Z. et al. mRNA therapeutics deliver a hopeful message. Nano Today23, 16–39. 10.1016/j.nantod.2018.10.005 (2018). - DOI
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

