Anti-Tumor Effects of Cecropin A and Drosocin Incorporated into Macrophage-like Cells Against Hematopoietic Tumors in Drosophila mxc Mutants
- PMID: 40136638
- PMCID: PMC11940895
- DOI: 10.3390/cells14060389
Anti-Tumor Effects of Cecropin A and Drosocin Incorporated into Macrophage-like Cells Against Hematopoietic Tumors in Drosophila mxc Mutants
Abstract
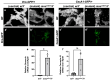
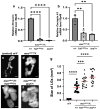
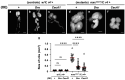
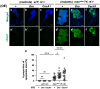
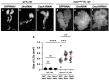
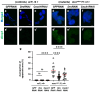
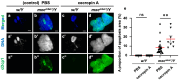
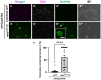
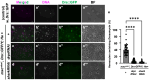
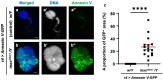
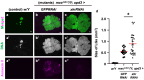
Five major antimicrobial peptides (AMPs) in Drosophila are induced in multiple sex combs (mxc) mutant larvae harboring lymph gland (LG) tumors, and they exhibit anti-tumor effects. The effects of other well-known AMPs, Cecropin A and Drosocin, remain unexplored. We investigated the tumor-elimination mechanism of these AMPs. A half-dose reduction in either the Toll or Imd gene reduced the induction of these AMPs and enhanced tumor growth in mxcmbn1 mutant larvae, indicating that their anti-tumor effects depend on the innate immune pathway. Overexpression of these AMPs in the fat body suppressed tumor growth without affecting cell proliferation. Apoptosis was promoted in the mutant but not in normal LGs. Conversely, knockdown of them inhibited apoptosis and enhanced tumor growth; therefore, they inhibit LG tumor growth by inducing apoptosis. The AMPs from the fat body were incorporated into the hemocytes of mutant but not normal larvae. Another AMP, Drosomycin, was taken up via phagocytosis factors. Enhanced phosphatidylserine signals were observed on the tumor surface. Inhibition of the signals exposed on the cell surface enhanced tumor growth. AMPs may target phosphatidylserine in tumors to induce apoptosis and execute their tumor-specific effects. AMPs could be beneficial anti-cancer drugs with minimal side effects for clinical development.
Keywords: Drosophila tumor; antimicrobial peptides (AMPs); apoptosis; endocytosis; hemocytes; phosphatidylserine.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

