Prolonged Low-Dose Administration of FDA-Approved Drugs for Non-Cancer Conditions: A Review of Potential Targets in Cancer Cells
- PMID: 40141362
- PMCID: PMC11942989
- DOI: 10.3390/ijms26062720
Prolonged Low-Dose Administration of FDA-Approved Drugs for Non-Cancer Conditions: A Review of Potential Targets in Cancer Cells
Abstract
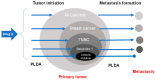
Though not specifically designed for cancer therapy, several FDA-approved drugs such as metformin, aspirin, and simvastatin have an effect in lowering the incidence of cancer. However, there is a great discrepancy between in vitro concentrations needed to eliminate cancer cells and the plasma concentration normally tolerated within the body. At present, there is no universal explanation for this discrepancy and several mechanisms have been proposed including targeting cancer stem cells (CSCs) or cellular senescence. CSCs are cells with the ability of self-renewal and differentiation known to be resistant to chemotherapy. Senescence is a response to damage and stress, characterized by permanent cell-cycle arrest and apoptotic resistance. Although, for both situations, there are few examples where low concentrations of the FDA-approved drugs were the most effective, there is no satisfactory data to support that either CSCs or cellular senescence are the target of these drugs. In this review, we concisely summarize the most used FDA-approved drugs for non-cancer conditions as well as their potential mechanisms of action in lowering cancer incidence. In addition, we propose that prolonged low-dose administration (PLDA) of specific FDA-approved drugs can be useful for effectively preventing metastasis formation in selected patients.
Keywords: carcinogenesis metastasis; chemoprevention; plasticity; senescence; stem cells.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures
Similar articles
-
Targeting of human cancer stem cells predicts efficacy and toxicity of FDA-approved oncology drugs.Cancer Lett. 2024 Sep 1;599:217108. doi: 10.1016/j.canlet.2024.217108. Epub 2024 Jul 8. Cancer Lett. 2024. PMID: 38986735
-
Novel anti-glioblastoma agents and therapeutic combinations identified from a collection of FDA approved drugs.J Transl Med. 2014 Jan 17;12:13. doi: 10.1186/1479-5876-12-13. J Transl Med. 2014. PMID: 24433351 Free PMC article.
-
Identification of FDA-approved drugs targeting breast cancer stem cells along with biomarkers of sensitivity.Sci Rep. 2013;3:2530. doi: 10.1038/srep02530. Sci Rep. 2013. PMID: 23982413 Free PMC article.
-
Can nanomedicines kill cancer stem cells?Adv Drug Deliv Rev. 2013 Nov;65(13-14):1763-83. doi: 10.1016/j.addr.2013.09.016. Epub 2013 Oct 10. Adv Drug Deliv Rev. 2013. PMID: 24120657 Free PMC article. Review.
-
Targeting Cancer Stem Cells with Repurposed Drugs to Improve Current Therapies.Recent Pat Anticancer Drug Discov. 2021;16(2):136-160. doi: 10.2174/1574892816666210208232251. Recent Pat Anticancer Drug Discov. 2021. PMID: 33563159 Review.
Cited by
-
The Seminal Role of the Proinflammatory Cytokine IL-1β and Its Signaling Cascade in Glioblastoma Pathogenesis and the Therapeutic Effect of Interleukin-1β Receptor Antagonist (IL-1RA) and Tolcapone.Int J Mol Sci. 2025 Jul 18;26(14):6893. doi: 10.3390/ijms26146893. Int J Mol Sci. 2025. PMID: 40725140 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

