A New Era of Muscarinic Acetylcholine Receptor Modulators in Neurological Diseases, Cancer and Drug Abuse
- PMID: 40143145
- PMCID: PMC11945405
- DOI: 10.3390/ph18030369
A New Era of Muscarinic Acetylcholine Receptor Modulators in Neurological Diseases, Cancer and Drug Abuse
Abstract
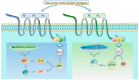
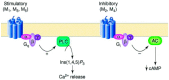
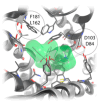
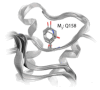
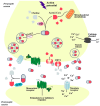
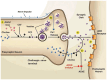
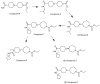
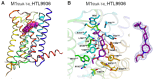
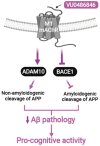
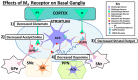
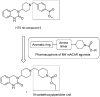
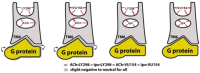
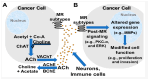
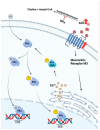
The cholinergic pathways in the central nervous system (CNS) play a pivotal role in different cognitive functions of the brain, such as memory and learning. This review takes a dive into the pharmacological side of this important part of CNS function, taking into consideration muscarinic receptors and cholinesterase enzymes. Targeting a specific subtype of five primary muscarinic receptor subtypes (M1-M5) through agonism or antagonism may benefit patients; thus, there is a great pharmaceutical research interest. Inhibition of AChE and BChE, orthosteric or allosteric, or partial agonism of M1 mAChR are correlated with Alzheimer's disease (AD) symptoms improvement. Agonism or antagonism on different muscarinic receptor subunits may lessen schizophrenia symptoms (especially positive allosteric modulation of M4 mAChR). Selective antagonism of M4 mAChR is a promising treatment for Parkinson's disease and dystonia, and the adverse effects are limited compared to inhibition of all five mAChR. Additionally, selective M5 antagonism plays a role in drug independence behavior. M3 mAChR overexpression is associated with malignancies, and M3R antagonists seem to have a therapeutic potential in cancer, while M1R and M2R inhibition leads to reduction of neoangiogenesis. Depending on the type of cancer, agonism of mAChR may promote cancer cell proliferation (as M3R agonism does) or protection against further tumor development (M1R agonism). Thus, there is an intense need to discover new potent compounds with specific action on muscarinic receptor subtypes. Chemical structures, chemical modification of function groups aiming at action enhancement, reduction of adverse effects, and optimization of Drug Metabolism and Pharmacokinetics (DMPK) will be further discussed, as well as protein-ligand docking.
Keywords: Alzheimer’s disease; CNS; cancer; cholinergic system; drug abuse; muscarinic receptors; schizophrenia.
Conflict of interest statement
The authors declare no conflict of interest.
Figures




Similar articles
-
Fine Tuning Muscarinic Acetylcholine Receptor Signaling Through Allostery and Bias.Front Pharmacol. 2021 Jan 29;11:606656. doi: 10.3389/fphar.2020.606656. eCollection 2020. Front Pharmacol. 2021. PMID: 33584282 Free PMC article. Review.
-
Structural Features of Iperoxo-BQCA Muscarinic Acetylcholine Receptor Hybrid Ligands Determining Subtype Selectivity and Efficacy.ACS Chem Neurosci. 2022 Jan 5;13(1):97-111. doi: 10.1021/acschemneuro.1c00572. Epub 2021 Dec 14. ACS Chem Neurosci. 2022. PMID: 34905693
-
Ligand bias at the muscarinic acetylcholine receptor family: Opportunities and challenges.Neuropharmacology. 2024 Nov 1;258:110092. doi: 10.1016/j.neuropharm.2024.110092. Epub 2024 Jul 25. Neuropharmacology. 2024. PMID: 39067666 Review.
-
Selective allosteric modulation of muscarinic acetylcholine receptors for the treatment of schizophrenia and substance use disorders.Adv Pharmacol. 2019;86:153-196. doi: 10.1016/bs.apha.2019.05.001. Epub 2019 Jul 22. Adv Pharmacol. 2019. PMID: 31378251 Review.
-
M1 muscarinic acetylcholine receptors: A therapeutic strategy for symptomatic and disease-modifying effects in Alzheimer's disease?Adv Pharmacol. 2020;88:277-310. doi: 10.1016/bs.apha.2019.12.003. Epub 2020 Jan 27. Adv Pharmacol. 2020. PMID: 32416870 Review.
References
-
- Boccardi M., Boccardi V. Factors Affecting Neurological Aging. Elsevier; Amsterdam, The Netherlands: 2021. Linking Aging, Anticholinergic Drugs Use, and Cognitive Impairment; pp. 453–461.
-
- Hampel H., Mesulam M.-M., Cuello A.C., Farlow M.R., Giacobini E., Grossberg G.T., Khachaturian A.S., Vergallo A., Cavedo E., Snyder P.J., et al. The Cholinergic System in the Pathophysiology and Treatment of Alzheimer’s Disease. Brain. 2018;141:1917–1933. doi: 10.1093/brain/awy132. - DOI - PMC - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

