Synthesis of Protoberberine Alkaloids by C-H Functionalization and Anionic Aza-6π-Electrocyclization: Dual Activity as AMPK Activators and Inhibitors
- PMID: 40151253
- PMCID: PMC11937996
- DOI: 10.1021/jacsau.5c00047
Synthesis of Protoberberine Alkaloids by C-H Functionalization and Anionic Aza-6π-Electrocyclization: Dual Activity as AMPK Activators and Inhibitors
Abstract
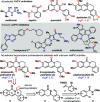
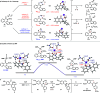
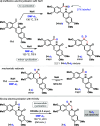
5'-Adenosine monophosphate-activated protein kinase (AMPK) plays a critical role in maintaining cellular energy homeostasis, and its activation has garnered attention for treating chronic metabolic diseases. Inhibitors of AMPK are underdeveloped but bear implications in treating cancers, controlling autophagy, and elderly wasting. Protoberberine alkaloids are typically regarded as AMPK activators. Herein, we report a modular synthesis strategy to access a collection of oxyberberine alkaloids, including the first synthesis of stepharotudine. In vitro assays reveal how subtle structural modifications can negate AMPK activation while conferring unprecedented inhibitory properties within the same class of compounds, which was previously unknown. Key steps in the synthesis include an oxidative Rh(III)-catalyzed C-H functionalization using electron-rich alkenes, NaH-mediated reductive N-O bond cleavage, and a rare example of an anionic aza-6π-electrocyclization. Additionally, we provide mechanistic support for nucleophilic hydride transfer reactivity with NaH in DMF.
© 2025 The Authors. Published by American Chemical Society.
Conflict of interest statement
The authors declare the following competing financial interest(s): The authors declare that the novel protoberberine-based AMPK inhibitors described in this study is included in a provisional patent that is jointly owned by the authors and the University of California, Riverside (UCR). The potential financial interest in the patent does not affect the integrity or objectivity of the study. All research findings and conclusions are independent of any financial and intellectual property considerations.
Figures



References
-
- Spaulding H. R.; Yan Z. AMPK and the Adaptation to Exercise. Annu. Rev. Physiol. 2022, 84, 209–227. 10.1146/annurev-physiol-060721-095517. - DOI - PMC - PubMed
- Steinber G. R.; Carling D. AMP-activated protein kinase: the current landscape for drug development. Nat. Rev. Drug Discovery 2019, 18, 527–551. 10.1038/s41573-019-0019-2. - DOI - PubMed
-
- Pollard A. E.; Martins L.; Muckett P. J.; Khadayate S.; Bornot A.; Clausen M.; Admyre T.; Bjursell M.; Fiadeira R.; Wilson L.; Whilding C.; Kotiadis V. N.; Duchen M. R.; Sutton D.; Penfold L.; Sardini A.; Bohlooly-Y M.; Smith D. M.; Read J. A.; Snowden M. A.; Woods A.; Carling D. AMPK activation protects against diet induced obesity through Ucp1-independent thermogenesis in subcutaneous white adipose tissue. Nat. Metab. 2019, 1, 340–349. 10.1038/s42255-019-0036-9. - DOI - PMC - PubMed
- Liu Y.; Jurczak M. J.; Lear T. B.; Lin B.; Larsen M. B.; Kennerdell J. R.; Chen Y.; Huckestein B. R.; Nguyen M. K.; Tuncer F.; Jiang Y.; Monga S. P.; O’Donnell C. P.; Finkel T.; Chen B. B.; Mallampalli R. K. A Fbxo48 inhibitor prevents pAMPKα degradation and ameliorates insulin resistance. Nat. Chem. Bio. 2021, 17, 298–306. 10.1038/s41589-020-00723-0. - DOI - PMC - PubMed
- Zhang E.; Jin L.; Wang Y.; Tu J.; Zheng R.; Ding L.; Fang Z.; Fan M.; Al-Abdullah I.; Natarajan R.; Ma K.; Wang Z.; Riggs A. D.; Shuck S. C.; Yang L.; Huang W. Intestinal AMPK modulation of microbiota mediates crosstalk with brown fat to control thermogenesis. Nat. Commun. 2022, 13, 1135. 10.1038/s41467-022-28743-5. - DOI - PMC - PubMed
- Steinberg G. R.; Hardie D. G. New insights into activation and function of the AMPK. Nat. Rev. Mol. Cell. Biol. 2023, 24, 255. 10.1038/s41580-022-00547-x. - DOI - PubMed
-
- Jeon S.-M.; Chandel N. S.; Hay N. AMPK regulates NADPH homeostasis to promote tumour cell survival during energy stress. Nature 2012, 485, 661–665. 10.1038/nature11066. - DOI - PMC - PubMed
- Jeon S.-M.; Hay N. The double-edged sword of AMPK signaling in cancer and its therapeutic implications. Arch. Pharm. Res. 2015, 38, 346–357. 10.1007/s12272-015-0549-z. - DOI - PMC - PubMed
- Dasgupta B.; Chhipa R. R. Evolving Lessons on the Complex Role of AMPK in Normal Physiology and Cancer. Trends Pharmacol. Sci. 2016, 37, 192–206. 10.1016/j.tips.2015.11.007. - DOI - PMC - PubMed
- Eichner L. J.; Brun S. N.; Herzig S.; Young N. P.; Curtis S. D.; Shackelford D. B.; Shokhirev M. N.; Leblanc M.; Vera L. I.; Hutchins A.; Ross D. S.; Shaw R. J.; Svensson R. U. Genetic Analysis Reveals AMPK Is Required to Support Tumor Growth in Murine Kras-Dependent Lung Cancer Models. Cell Metab. 2019, 29, 285–302.e7. 10.1016/j.cmet.2018.10.005. - DOI - PMC - PubMed
- Sadria M.; Seo D.; Layton A. T. The mixed blessing of AMPK signaling in Cancer treatments. BMC Cancer 2022, 22, 105. 10.1186/s12885-022-09211-1. - DOI - PMC - PubMed
-
- Whitmarsh-Everiss T.; Laraia L. Small molecule probes for targeting autophagy. Nat. Chem. Biol. 2021, 17, 653–664. 10.1038/s41589-021-00768-9. - DOI - PubMed
- Barnaba C.; Broadbent D. G.; Kaminsky E. G.; Perez G. I.; Schmidt J. C. AMPK regulates phagophore-to-autophagosome maturation. J. Cell. Biol. 2024, 223, e202309145 10.1083/jcb.202309145. - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
