A real-life evaluation of SNOT-22 domains in a cohort of CRSwNP patients treated with biologic therapies for 12 months
- PMID: 40151546
- PMCID: PMC11946757
- DOI: 10.1016/j.waojou.2025.101041
A real-life evaluation of SNOT-22 domains in a cohort of CRSwNP patients treated with biologic therapies for 12 months
Abstract
Background: Chronic rhinosinusitis with nasal polyps (CRSwNP) is an inflammatory disorder associated with rhinorrhea, nasal obstruction, nasal congestion, hyposmia, anosmia, and facial pain or pressure for over 12 weeks. This study examines the Sino-Nasal Outcome Test 22 (SNOT-22) score and its relationship to nasal, otologyc, sleep and emotional domains in CRSwNP patients during the first year of biologics treatment, comparing the pre-biologics score to that at 1, 6, and 12 months in a cohort of 59 patients with CRSwNP.
Methods: We included 59 patients with CRSwNP (with or without asthma) who received add on therapy with targeted monoclonal antibodies (mAbs). At each visit we administered the SNOT-22 questionnaire and both total score and single domains scores were recorded.
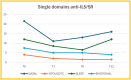
Results: In this real-life, observational study, we found a significant SNOT-22 total score reduction for patients treated with anti-IgE after 1 month, but this significant difference was not maintained at 6 or 12 months compared with the baseline. The use of anti-interleukin 5/5R (IL5/5R) leads to a significant reduction of the SNOT-22 total score after 1 month, which is maintained after 6 months but not at 12 months compared with the baseline. The use of an anti-interleukin 13/4R (IL13/4R) leads to a statistically significant reduction of the SNOT-22 score after 1 month of therapy, which is maintained after 6 and 12 months compared with the baseline. When examining the single domains, we observed that patients who received anti-IL13/4R treatment demonstrated a significant reduction in each domain at each time point (T) compared to the baseline. Patients who received anti-IL5/5R treatment demonstrated an improvement in the nasal domain at each T compared to the baseline. However, the improvement in the otologyc domain was not sustained after 12 months. Similarly, the sleep domain remained unchanged, and the emotional domain only improved significantly after 12 months. Similarly, there was a reduction of the emotional domain in patients treated with anti-IgE.
Conclusion: Our real-life study describes the kinetics over the first year of treatment with mAbs in CRSwNP, showing different patterns in reducing symptoms and improving Health Related Quality of Life (HRQoL). SNOT-22 with the factorial division in 4 domains can help distinguish fast responders from low or non-responders to a mAb based on clinical response after 1 month and more accurately assign the right mAb to the right patient.
Keywords: Asthma; Biologicals; CRSwNP; Monoclonal antibodies; SNOT-22.
© 2025 The Authors.
Conflict of interest statement
GAML. Costanzo declares no competing interests, AG. Ledda declares no competing interests, G. Sambugaro declares no competing interests, G. Murdaca declares no competing interests, C. Caruso declares no competing interests, S. Canalis declares no competing interests, P. Serra declares no competing interests, MP. Barca declares no competing interests, S. Del Giacco has received speaker fees by AstraZeneca, Chiesi, GSK, Guidotti, Menarini, Novartis, Sanofi, Stallergenes, Takeda and Valeas. He has received advisory board fees from AstraZeneca, Chiesi, CSL-Behring, GSK, Novartis, Sanofi and Takeda, and has received research grants from AstraZeneca, CSL.Behring, GSK and Novartis. D. Firinu declare no competing interests.
Figures


References
-
- Bachert C., Han J.K., Desrosiers M., et al. Efficacy and safety of dupilumab in patients with severe chronic rhinosinusitis with nasal polyps (LIBERTY NP SINUS-24 and LIBERTY NP SINUS-52): results from two multicentre, randomised, double-blind, placebo-controlled, parallel-group phase 3 trials. Lancet. 2019;394(10209):1638–1650. doi: 10.1016/S0140-6736(19)31881-1. - DOI - PubMed
LinkOut - more resources
Full Text Sources

